By Madalina Diaconu, Product Manager Pretect D, and Dr. Ajay Awati, Global Category Manager Gut Health & Nutrition, EW Nutrition
Coccidiosis is one of the most devastating enteric challenge in the poultry industry costing over over 14 billion US$ per year (Blake et al., 2020). In the early days of intensive poultry production, outbreaks of Eimeria tenella, were most destructive. Eimeria tenella is a coccidia species that causes severe haemorrhages and hypovolemic shock, leading to a fatal outcome for the affected bird.
 Poultry producers need to control the performance and welfare issues caused by subclinical coccidiosis
Poultry producers need to control the performance and welfare issues caused by subclinical coccidiosis
Understanding and managing coccidiosis in poultry
However, today, subclinical coccidiosis accounts for even more of production losses due to intestinal cells injuries: lower body weights, higher feed conversion rates, lack of flock uniformity, failures on skin pigmentation and, at the end mortality. Variation in the supply and quality of animal feed exacerbates the issue and compromises farm profitability even more. To tackle this challenge, we need to understand the basics of coccidiosis control in poultry and what options producers have to manage coccidiosis risks.
From Eimeria infection to disease
Coccidiosis is a disease caused by protozoan parasites, mainly of the genus Eimeria, that are located in the small and large intestines. Being very resistant and highly contagious, these protozoa are easily transmitted by various routes (via feed, litter, water, soil, material, insects, and wild animals).
Coccidia are present in all livestock species. However, the infection is particularly severe in poultry. The health consequences can be significant: loss of appetite, reduction in feed intake, increased FCR, enteritis, hemorrhagic diarrhea, and mortality. The most common species of Eimeria in broilers are: E. acervulina, E. mitis, E. maxima, E. brunetti, E. necatrix, E. praecox, and E. tenella. They are widely found in broiler productions across the globe (McDougall & Reid, 1991).
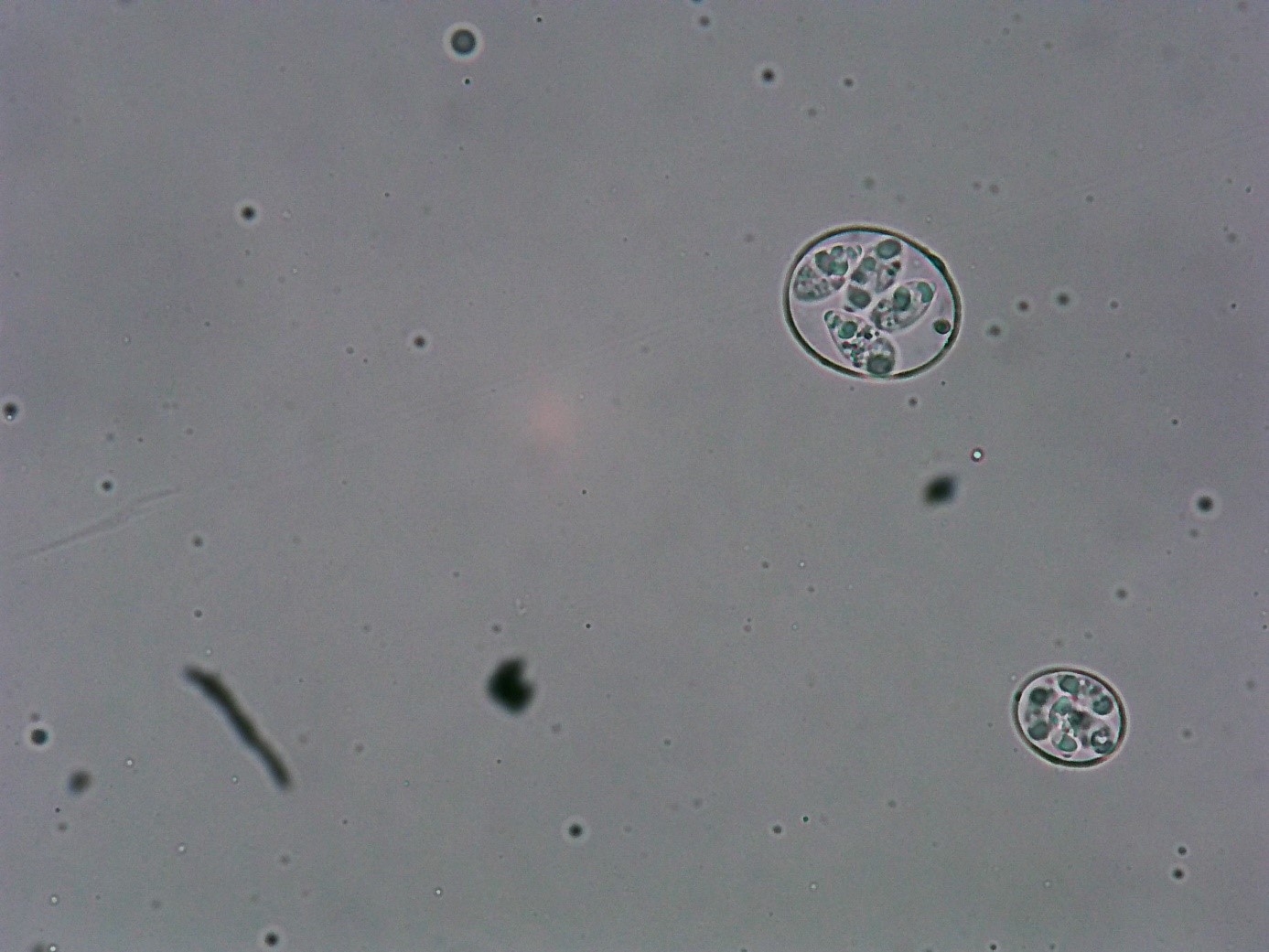 Figure 1: Sporulated oocyst of Eimeria maxima and E. Acervulina (40 x)
Figure 1: Sporulated oocyst of Eimeria maxima and E. Acervulina (40 x)
The pathogenesis of infection varies from mild to severe and is largely dependent on the magnitude of infection. Coccidiosis outbreaks are related to multiple factors that, together, promote a severe infestation in the farm.
Within poultry, the highest economic impact is in broilers, where the most common species of Eimeria are E. acervulina, E. maxima, E. tenella and E. necatrix, which all show high virulence. However, pathogenicity is influenced by host genetics, nutritional factors, concurrent diseases, age of the host and the particular species of the Eimeria (Conway & McKenzie, 2007).
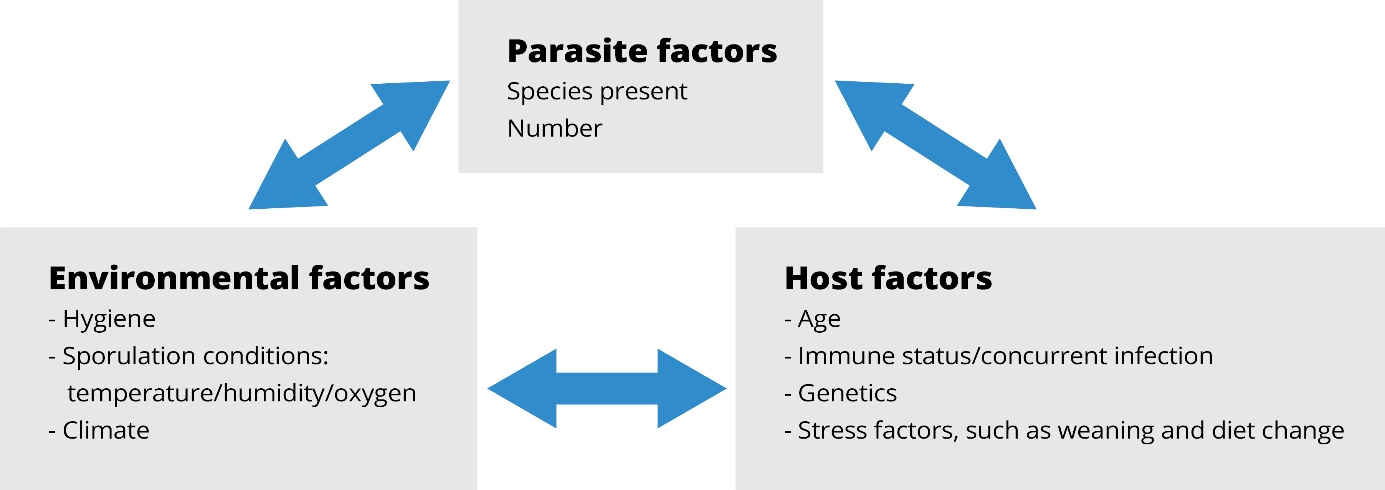 Figure 2: Interaction of factors that promote coccidia outbreaks
Figure 2: Interaction of factors that promote coccidia outbreaks
The Eimeria infection starts with the ingestion of protozoa that are at a sporulated stage. Once inside the gut, the protozoa liberate the sporozoites. This infective form can get into enterocytes and then begin a massive reproduction, killing thousands of intestinal cells. (Olabode et al., 2020; Shivaramaiah et al., 2014)
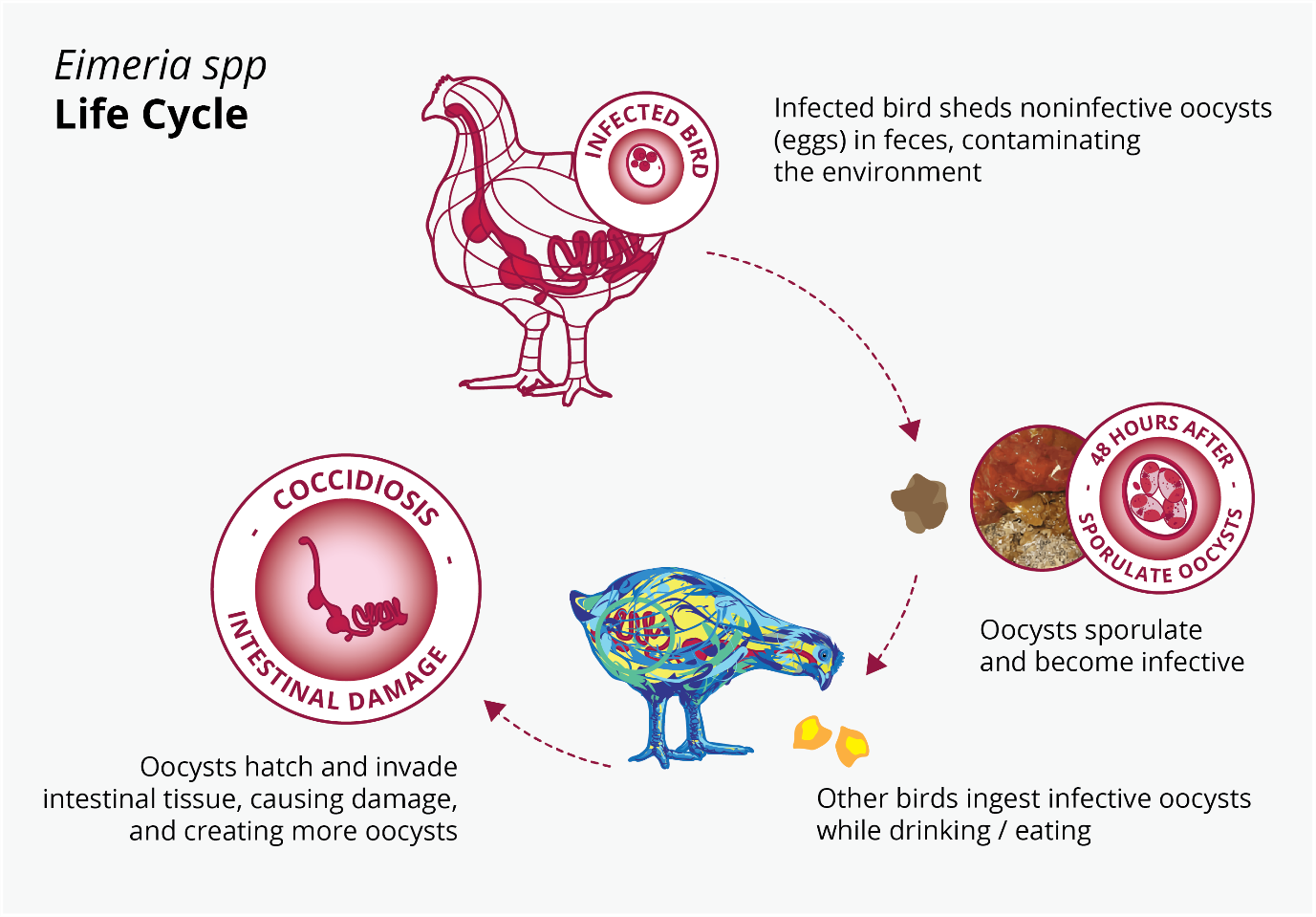 Figure 3: Eimeria spp. life cycle
Figure 3: Eimeria spp. life cycle
The reproduction potential depends on the coccidia species. E. acervulina, E. mitis and E. praecox have the highest reproduction rate. This characteristic is closely related to their short life cycle.
In broilers, coccidiosis usually occurs after 21 days of age. The infection spreads gradually from day 1 already, depending on species of Eimeria and their virulence. A typical progression of coccidiosis in broilers is shown in Figure 4.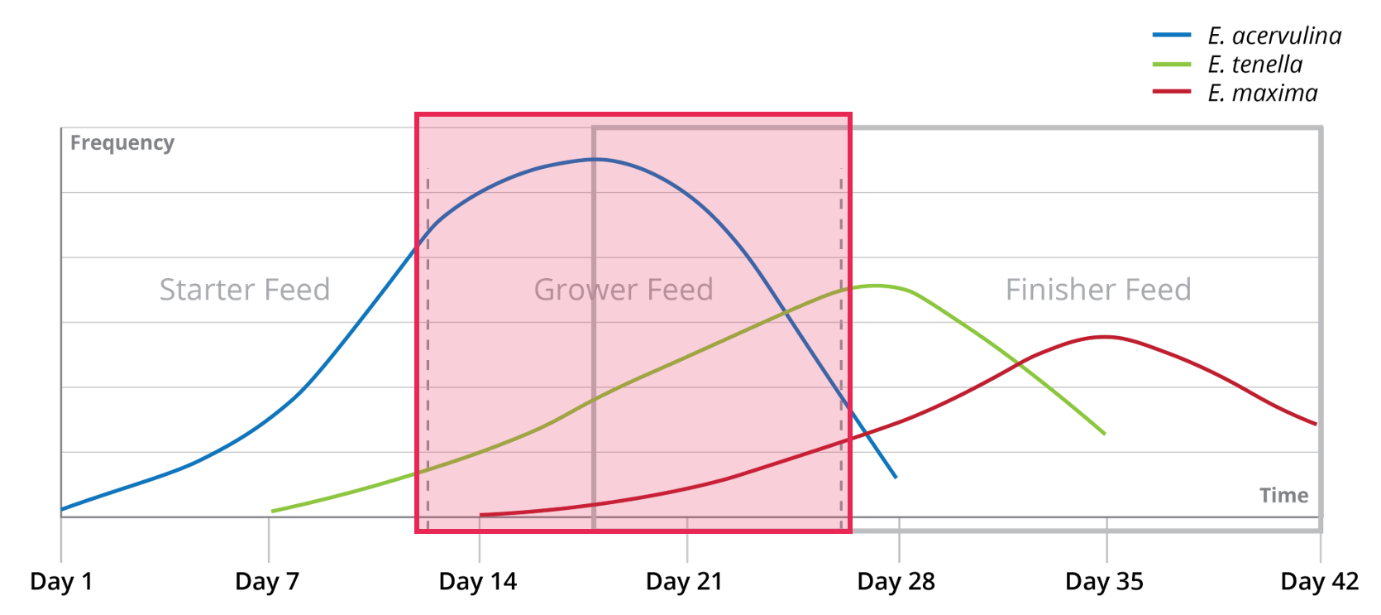 Figure 4: Typical development of a coccidia infection in relation to broiler feed phases
Figure 4: Typical development of a coccidia infection in relation to broiler feed phases
Coccidiosis control in poultry: Strategy guidelines
The intrinsic characteristics of coccidiosis makes this parasite unique and many times frustrating to control. Resistance to available coccidiostats makes this task even harder. Good farm management, litter hygiene, and the use of control coccidiosis programs such as shuttle and rotation are functional measures to prevent clinical coccidiosis. Successful control strategies specifically recognize the importance of monitoring, use anticoccidial drugs wisely, and include vaccines where applicable.
Monitoring
The first step is to establish a strict monitoring program in all stages of production, including the feed mill. It is important to verify that therapeutics are included in the feed in an adequate form and quantity, and that the follow-up in the field takes place.
Field monitoring should be frequent and in line with the operation’s coccidiosis management program. Field monitoring is a complementary work that collates clinical, necropsy, and faeces findings to closely track the disease situation.
 Coccidiosis control in poultry operations needs to include rigorous monitoring
Coccidiosis control in poultry operations needs to include rigorous monitoring
Anticoccidial drugs
Since the middle of the 20th century, chemotherapeutic agents have offered the best way to control coccidia. However, unbridled use of anti-coccidial drugs and the emergence of the new resistant field strains of coccidia have made it increasingly challenging to control coccidiosis with commonly available coccidiostat drugs.
The coccidiostats have been classified in two groups: ionophores, molecules obtained from microbiological fermentation, and chemicals, synthetic compounds. The mode of action of ionophores is to interfere with the membrane ion exchange, killing the extracellular stages (sporozoites and or merozoites) as they expend energy to maintain the osmotic balance. Chemical compounds can have an anticoccidial effect even on extracellular and intracellular stages (Sumano López & Gutiérrez Olvera, 2005).
However, resistance development is limiting their effectiveness, and certain compounds cannot be used in older birds or in hot environments. Moreover, government regulations often include anti-coccidial drugs in bans on antibiotics use. This does not mean that these drugs are not crucial to controlling this disease, but it is important to use alternative tools: they help make a coccidiosis control program not only less dependent on anticoccidial drugs but also more robust.
Vaccines
There are two commercial kinds of coccidia vaccines; the first one uses natural strains. These Eimeria are selected from field outbreaks, show a medium pathogenicity, and allow for a controlled replication of a coccidia infection. The second kind of vaccines include attenuated strains; these are precocious strains and birds usually show low or no post-vaccinal reactions.
The management of coccidia vaccines is the principal challenge for using this tool to control coccidia. Special vaccination training is required at the hatchery, which then needs a follow-up on the farm. In the field, this follow-up and the alignment of all the protocols has proven challenging for many producers.
Managing coccidiosis in poultry: Next steps
The limitations chemotherapy and vaccines have led to a surge in the quest for effective natural solutions. Recent research into plant-derived phytochemicals shows that these compounds have properties that make them an interesting tool against coccidiosis (cf. Cobaxin-Cárdenas, 2018). Knowledge, research, and technological developments are now ready to offer solutions that can be an effective part of coccidia control programs. These natural solutions create opportunities to make poultry production more sustainable by reducing dependency on harmful drugs.
References
Bafundo, K.W., L. Gomez, B. Lumpkins, G.F. Mathis, J.L. McNaughton, and I. Duerr. “Concurrent Use of Saponins and Live Coccidiosis Vaccines: The Influence of a Quillaja and Yucca Combination on Anticoccidial Effects and Performance Results of Coccidia-Vaccinated Broilers.” Poultry Science 100, no. 3 (2021): 100905. https://doi.org/10.1016/j.psj.2020.12.010.
Blake, Damer P., Jolene Knox, Ben Dehaeck, Ben Huntington, Thilak Rathinam, Venu Ravipati, Simeon Ayoade, et al. “Re-Calculating the Cost of Coccidiosis in Chickens.” Veterinary Research 51, no. 1 (September 14, 2020). https://doi.org/10.1186/s13567-020-00837-2.
Cobaxin-Cárdenas, Mayra E. “Natural Compounds as an Alternative to Control Farm Diseases: Avian Coccidiosis.” Farm Animals Diseases, Recent Omic Trends and New Strategies of Treatment, March 21, 2018. https://doi.org/10.5772/intechopen.72638.
Conway, Donal P., and M. Elizabeth McKenzie. Poultry Coccidiosis: Diagnostic and Testing Procedures. Ames, IA, IA: Blackwell Publishing, 2007.
McDougall, L. R., and W. M. Reid. “Coccidiosis.” Chapter. In Diseases of Poultry, edited by B. W. Calnek, H. W. Yoder, W. M. Reid, C. W. Beard, and H. J. Barnes. Ames, IA: Iowa State University Press, 1991.
Olabode, Victoria Bose, Dashe Yakubu Gunya, Umaru Mada Alsea, Tobias Peter Pwajok Choji, and Israel Joshua Barde. 2020. “Histopathological Lesions of Coccidiosis Natural Infestation in Chickens”. Asian Journal of Research in Animal and Veterinary Sciences 5 (2), 41-45. https://www.journalajravs.com/index.php/AJRAVS/article/view/30090.
Shivaramaiah, Chaitanya, John R. Barta, Xochitl Hernandez-Velasco, Guillermo Téllez, and Billy M. Hargis. “Coccidiosis: Recent Advancements in the Immunobiology of Eimeria Species, Preventive Measures, and the Importance of Vaccination as a Control Tool against These Apicomplexan Parasites.” Veterinary Medicine: Research and Reports 2014, no. 5 (April 28, 2014): 23–34. https://doi.org/10.2147/vmrr.s57839.
Sumano López, Héctor, and Gutiérrez Olvera Lilia. Farmacología Clínica En Aves Comerciales. México: UNAM, Departamento de Fisiología y Farmacología, 2005.















