Diseases caused by E. coli entail use of antibiotics in animal production
E. coli infections are a major problem in pig production. Especially young animals with an incompletely developed immune system are often unable to cope with the cavalcade of pathogens. In poultry, E. coli are responsible for oedema, but also for respiratory diseases. In young piglets, E. coli cause diarrhoea , oedema, endotoxic shock and death. In order to cure the animals, antibiotics often must be applied. Besides this curative application, antibiotics were and in many countries still are used prophylactically and as growth promoters.
The excessive use of antibiotics, however, leads to the occurrence of antimicrobial resistance (AMR): due to mutations, resistance genes are created which enable enterobacteria such as Salmonella, Klebsiella and E. coli to produce enzymes (ß-lactamases) in order to withstand ß-lactam antibiotics. In case of an antibiotic treatment, the resistant bacteria survive whereas the other bacteria die.
The major problem here is that these resistance genes can be transferred to other bacteria. Harmless bacteria can thus transfer resistance genes to dangerous pathogens, which then cannot be combatted with antibiotics anymore. In this article we explore in detail how AMR happens and how phytomolecules, which have antimicrobial properties, could be a key tool to reduce the need for antibiotics in animal production.
How ß-lactam antibiotics work
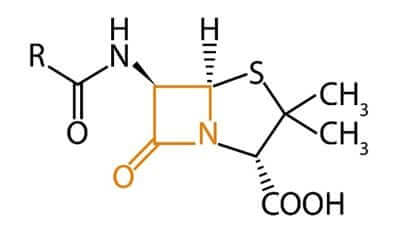
The group of ß-lactam antibiotics consists of penicillins, cephalosporins, monobactams, and carbapenems. These antibiotics are characterised by their lactam ring (Figure 1).
Figure 1: An antibiotic with a ß-lactam ring (in orange)
If bacteria are growing, the cell wall also has to grow. For this purpose existing conjunctions are cracked and new components are inserted. In order for the cell wall to remain a solid barrier, the new components must be interconnected by crosslinks. For the creation of these crosslinks an enzyme is essential, the transpeptidase (figure 2).
Figure 2: building up a stable cell wall with the help of transpeptidase
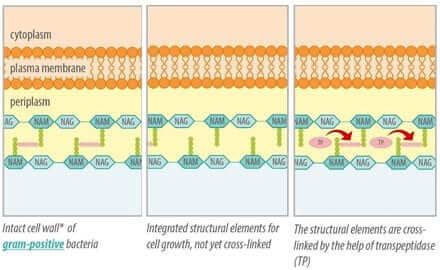
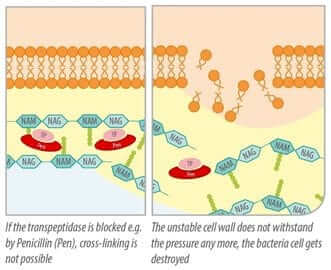
Due to their structure, ß-lactam-antibiotics also fit as binding partner for transpeptidase. They bind to the enzyme and block it (Kohanski et al., 2010). The crosslinks cannot be created and the stabilization of the cell wall is prevented. Disturbance of cell wall stability leads to the death of the bacterial cell, hence ß-lactam antibiotics act bactericidal.
Figure 3: blocked by ß-lactam antibiotics, transpeptidase cannot serve as enzyme for building the cell wall
The challenge: E. coli producing ß-lactamases
Resistant bacteria, which are able to produce ß-lactamases – enzymes that destroy the ß-lactam ring – prevent their own destruction. Divers point mutations within the ß-lactamase genes lead to the occurrence of “extended-spectrum-beta-lactamases“ (ESBL). ESBL are able to inactivate most of the ß-Lactam-antibiotics.
Another mutation leads to so-called AmpC (aminopenicillin and cephalosporin) ß-lactamases. They enable the E. coli to express a resistance against penicillins, cephalosporins of the second and third generation as well as against cephamycins.
Phytomolecules – an alternative?
One approach to reduce the use of antibiotics is the utilization of phytomolecules. These secondary metabolites are produced by plants to protect themselves from moulds, yeasts, bacteria and other harmful organisms.
The use of plants and their extracts in human and veterinary medicine is well-established for centuries. Besides digestive and antioxidant characteristics they are well known for their bacteriostatic and bactericidal effects.
Consisting of a high number of chemical compounds, they attack at diverse points and their antimicrobial effect is not caused by only one single specific mechanism. This is crucial because it is therefore very unlikely that bacteria can develop resistances to phytomolecules like they do to antibiotics.
How phytomolecules work
Mostly, phytomolecules act at the cell wall and the cytoplasm membrane level. Sometimes they change the whole morphology of the cell. This mode of action has been studied extensively for thymol and carvacrol, the major components of the oils of thyme and oregano.
They are able to incorporate into the bacterial membrane and to disrupt its integrity. This increases the permeability of the cell membrane for ions and other small molecules such as the energy carrier ATP (Adenosin-tri-phosphate). It leads to the decrease of the electrochemical gradient above the cell membrane and to the loss of energy equivalents of the cell.
A special challenge: gram-negative bacteria
Gram-negative bacteria such as E. coli and Salmonella pose a special challenge. The presence of lipopolysaccharides in the outer membrane (OM) provides the gram-negative bacteria with a hydrophilic surface (Nikaido, 2003; Nazarro et al., 2013) (see also blue infobox).
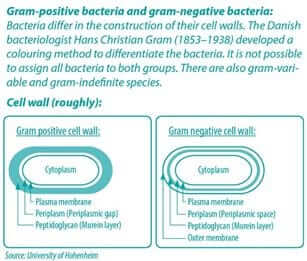
The cell wall therefore only allows the passage of small hydrophilic solutes and is a barrier against macromolecules and hydrophobic compounds such as hydrophobic antibiotics and toxic drugs. The bypassing of the OM therefore is a prerequisite for any solute to exert bactericidal activity toward gram-negative bacteria (Helander et al., 1998).
Based on their trial results Helander et al. (1998) (1998) concluded that trans-cinnamaldehyde and partly also thymol and carvacrol gain access to the periplasm and to the deeper parts of the cell. Nikaido (1996) also concluded that OM-traversing porin proteins allow the penetration of lipophilic probes at significant rates.
Evaluating phytomolecules I – in vitro trial, Scotland
A trial conducted in Scotland evaluated the effects of Activo Liquid, a mixture of selected phytomolecules and citric acid, on ESBL-producing E. coli as well as on E. coli that generate AmpC.
Material and methods
For the trial two strains for each group were isolated from the field, a non-resistant strain of E. coli served as control. Suspensions of the strains with 1×104 CFU/ml were incubated for 6-7 h at 37°C (98.6°F) together with diverse concentrations of Activo Liquid or with cefotaxime, a cephalosporin. The cefotaxime group saved as a control for differentiating resistant and non-resistant E. coli.
The suspensions were put on LB agar plates and bacteria colonies were counted after further 18-22h incubation at 37°C.
Results
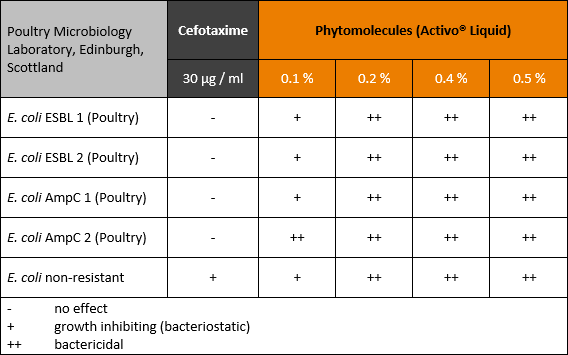
The antimicrobial efficacy of the blend of phytomolecules depended on the concentration at which they were used (see table 1). A bacteriostatic effect could be shown at dilutions up to 0.1 %, a bactericidal effect at higher concentrations.
Table 1: Effect of phytomolecules against resistant E. coli producing ESBL and AmpC in poultry
Evaluating phytomolecules II – in vitro trial, Germany
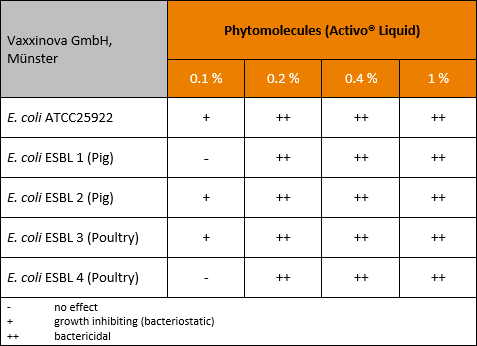
A further trial was conducted in Germany (Vaxxinova, Münster), confirming the preceding results.
Material and methods
Four ESBL producing E. coli all isolated from farms and a non-resistant reference strain as control were tested concerning their sensitivity against Activo Liquid. Every bacteria strain (Conc.:1×104 CFU/ml) was subjected to a bacterial inhibition assay in an appropriate medium at 37°C for 6-7 hours.
Results
In this trial Activo Liquid also showed a dose-dependent efficacy, with no or just a bacteriostatic effect up to a concentration of 0.1 %, but bactericidal effects at a concentration of ≥0.2 % (table 2).
Table 2: Effect of phytomolecules against resistant ESBL producing E. coli in pig and in poultry
Phytomolecules: a promising outlook
E. coli infections have devastating effects on animals, from diarrhea to edema, enterotoxic shock and even death. Antibiotic treatments have long been the only practicable answer. However, their excessive use ̶ for instance, the metaphylactic application to thousands of animals in a flock ̶ has led to the development of resistant strains. There is evidence that a reduction of antibiotic use reduces the occurrence of resistances (Dutil et al., 2010).
The results of the two in vitro trials in Scotland and Germany demonstrate the bactericidal effects of phytomolecules on E. coli that produce ESBL and AmpC. Using phytomolecules could thus reduce the use of antibiotics and therefore also the occurrence of AMR.
While it is theoretically possible for bacteria to also become resistant against phytomolecules, the probability of this happening is very low: unlike antibiotics, phytomolecules contain hundreds of chemical components with different modes of action. This makes it exceedingly difficult for bacteria to adapt and develop resistance. To tackle the problem of antibiotic-resistant E. coli, antimicrobial phytomolecules therefore offer a promising, sustainable and long-term solution.
By Dr. Inge Heinzl, Editor, EW Nutrition
Literature
Dutil, Lucie, Rebecca Irwin, Rita Finley, Lai King Ng, Brent Avery, Patrick Boerlin, Anne-Marie Bourgault, Linda Cole, Danielle Daignault, Andrea Desruisseau, Walter Demczuk, Linda Hoang, Greg B. Horsman, Johanne Ismail, Frances Jamieson, Anne Maki, Ana Pacagnella, and Dylan R. Pillai. 2010.” Ceftiofur Resistance in Salmonella enterica Serovar Heidelberg from Chicken Meat and Humans, Canada.” Emerg Infect Dis 16 (1): 48-54.
Helander, Ilkka M., Hanna-Leena Alakomi, Kyösti Latva-Kala, Tiina Mattila-Sandholm, Irene Pol, Eddy J. Smid, Leon G. M. Gorris, and Atte von Wright. 1998. “Characterization of the Action of Selected Essential Oil Components on Gram-Negative Bacteria.” J. Agric. Food Chem 46: 3590-595.
Kohanski, Michael A., Daniel J. Dwyer, and James J. Collins. 2010. “How Antibiotics Kill Bacteria: From Targets to Networks.” Nature Reviews 8: 423-435.
Nazarro, Filomena, Florinda Fratianni, Laura De Martino, Raffaele Coppola, and Vincenzo De Feo. 2013. “Effect of Essential Oils on Pathogenic Bacteria.” Pharmaceuticals 6 (12): 1451-1474.
Nikaido, Hiroshi ” Molecular Basis of Bacterial Outer Membrane Permeability Revisited. 2003. ” Microbiology and Molecular Biology Reviews, 67 (4): 593-656.
Rodriguez, Tori. 2015 “Essential Oils Might Be the New Antibiotics.” The Atlantic.
Rüben, Christiane. 2009. “Antimikrobielle Wirksamkeit von chemischen Einzelkomponenten ätherischer Öle gegenüber ausgewählten Lebensmittelverderbniserregern“. PhD diss, TeHo Hannover.