By Dr. Inge Heinzl, Editor, EW Nutrition
According to the American Medical Association, antimicrobial resistance is one of the main threats to public health nowadays. More than 2 million people are infected with bacteria resistant to different types of antibiotics every year (Marquardt and Suzhen, 2018). Prof Dame Sally Davies (2012), Chief Medical Officer for England, mentions that antibiotics are losing their effectiveness at alarming rates. Bacteria are finding ways to survive the antibiotics, so these molecules no longer work. O’Neill (2016) predicted in his report that 10 million people a year could be dying by 2050 due to antimicrobial resistance.
Antimicrobial resistance is a natural process but this is accelerated by inappropriate prescribing of antimicrobials, poor infection control practices and the unnecessary use of antimicrobials in agriculture (Barber and Sutherland, 2017).
Antimicrobial resistance – a threat to humanity
Resistance to specific antibiotics occurs through mutations that enable the bacteria to withstand an antibiotic treatment. One mechanism is the production of enzymes degrading or altering the antibiotic, rendering them harmless. The elimination of entrances for antibiotics or the development of pumps discharging them is another possibility. A further option is the elimination of the targets the antibiotic would attack.
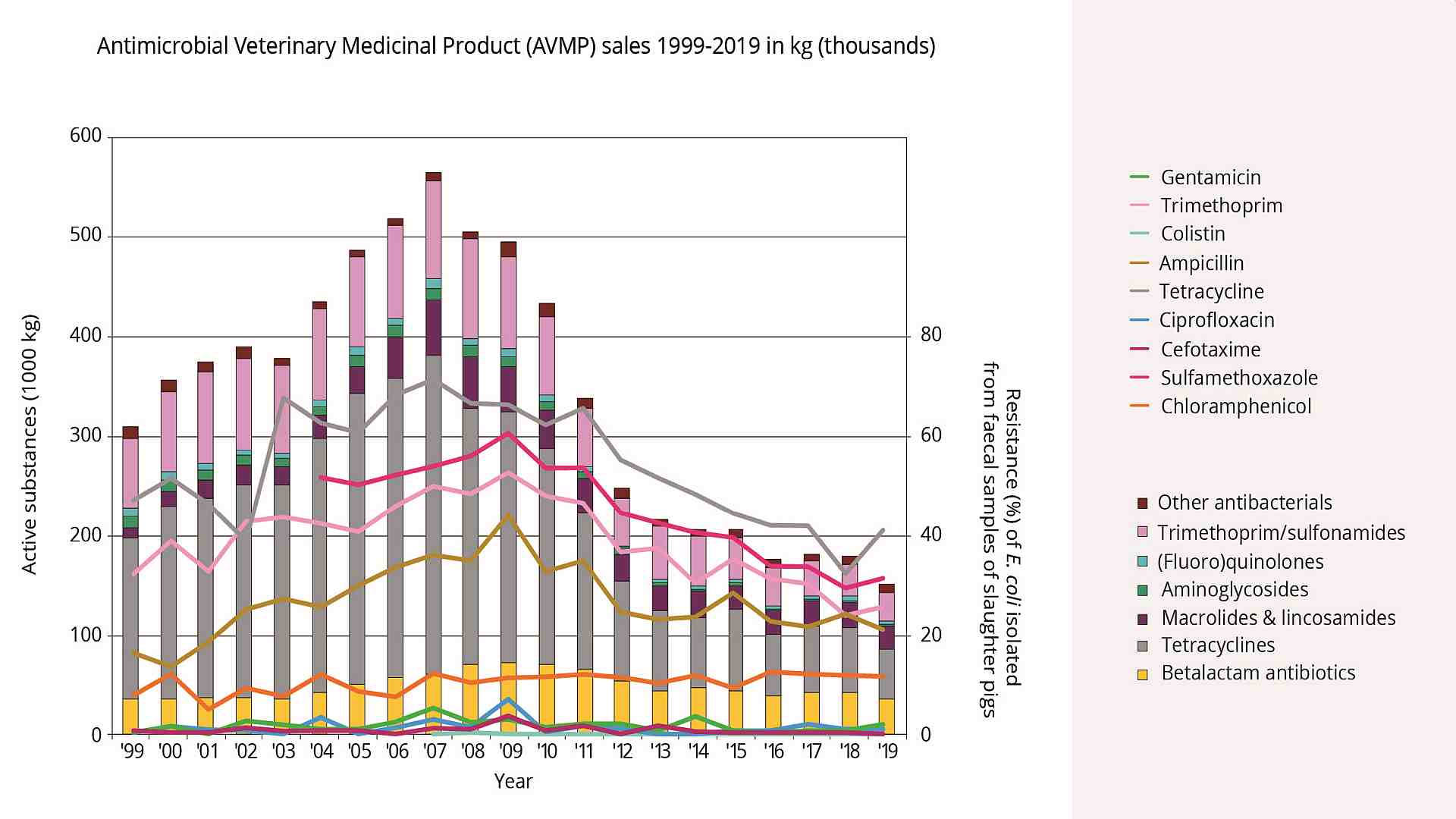
So-called “resistance genes” are responsible for resistance. These genes can be transferred from one bacterium to another and also from beneficial bacteria to harmful ones. When antibiotics are used, “normal” bacteria are killed; the resistant ones survive and have all possibilities to proliferate. The Dutch Government has been tracking resistant bacteria in poultry flocks for the last two decades. A clear correlation between antibiotic use and the percentage of resistance could be observed. The good thing: according to the 2020 MARAN report (De Greeff et al., 2020), by reducing the use of antibiotics, the occurrence of resistances can be pushed back.
Figure 1. Sales of antibiotics from 1999 to 2016 and the development of resistances (MARAN report, 2018)
Antibiotic use in animal production
In pig production, antibiotics are often used in stressful situations such as weaning or moving. Antibiotics decrease the pathogenic pressure in animals and help them overcome these critical periods. Disadvantage: Antibiotics do not differentiate between good and bad but between susceptible and resistant. Therefore, also the beneficial gut flora gets destroyed through antibiotic treatment, and resistance is spread.
After the ban of antibiotic growth promoters in Europe in 2006, the US has also made considerable efforts to reduce the use of antibiotics.
Is performance at risk without antibiotics?
When antibiotics are taken out of livestock production, measures in different areas must be implemented to keep performance and profitability high. Without supporting the animals by other means, they will get sick and even die in acute cases. Subclinical disease forms reduce their feed intake, and growth performance consequently decreases. According to literature, losses due to decreased average weight gain can be up to $40 per pig (Hao et al., 2014).
Goal: reducing antibiotics while maintaining performance
To support pigs, especially during the afore-mentioned critical periods, alternatives focusing on the maintenance of gut health and, therefore, also overall health must be chosen. This goal can only be achieved by balancing the intestinal flora with reducing pathogenic bacteria occurrence.
Phytomolecules are an effective solution
Phytomolecules are produced by plants to defend themselves against predators or pathogens. Farmers use the substances in animal feeds to support digestion, improve palatability, but also to reduce pathogenic pressure (Baser and Buchbauer, 2010).
In animal feeding, different application forms are available:
- As premixes containing microencapsulated phytomolecules with a slow release. This version is mixed into the feed in the feed mill and constitutes continuous long-term support for the animals. Due to microencapsulation, the active substances are released where they are needed – in the gut.
- As liquid complementary feeds for the application via the waterline. The application of the liquid form to the animals can be decided from one day to the other. It is an optimal additional tool to support the pigs in challenging situations such as weaning.
Scientific trials show: In-feed phytomolecules support performance
A trial conducted at the Federal University of Lavras (Brazil) evaluated if phytomolecules as a regular diet component can deliver the same effects on growth performance as AGPs in pig production.
For the trial, 108 castrated newborn male pigs were allocated to 3 groups (control, AGP (antibiotic growth promoters), and Activo). Pigs were weaned at 23 days of age with an average weight of 6.3 kg. They were fed a 3-phase diet (nursery, growing, and finishing). The inclusion rates of the additives (antibiotics and phytomolecules-based product – Activo) are shown in table 1.
On days 0, 1, and 2 of the experiment, the animals were challenged by applying a solution containing 107 CFU of E. coli K88, producing the toxins LT, Sta, and bST. Additionally, during the two last days before the growing phase, the animals were exposed to 5h of heat stress, using infrared lamps and closed windows. The parameters weight gain, final weight, FCR, and gut flora composition in the cecum were evaluated.
| Phase | Control | AGP | Activo | |
| Nursery | 0-7 days | — | Gentamycin 2.7kg/t | 0.4kg/t |
| 8-42 days | — | Haloquinol 0.2kg/t | 0.3kg/t | |
| Growing | 42-52 days | — | Tylosin 0.45kg/t | 0.4kg/t |
| 53-87 days | — | Enramycin 0.125kg/t | 0.2kg/t | |
| Finishing | 88-97 days | — | Tylosin 0.45kg/t | 0.4kg/t |
| 98-126 days | — | Enramycin 0.063kg/t | 0.2kg/t | |
Table 1. Inclusion rate of the additives in the feed
AGP: Antibiotic growth promoter; Activo: product based on phytomolecules, microencapsulated (EW Nutrition)
Results
The results of this trial are shown in figure 2.
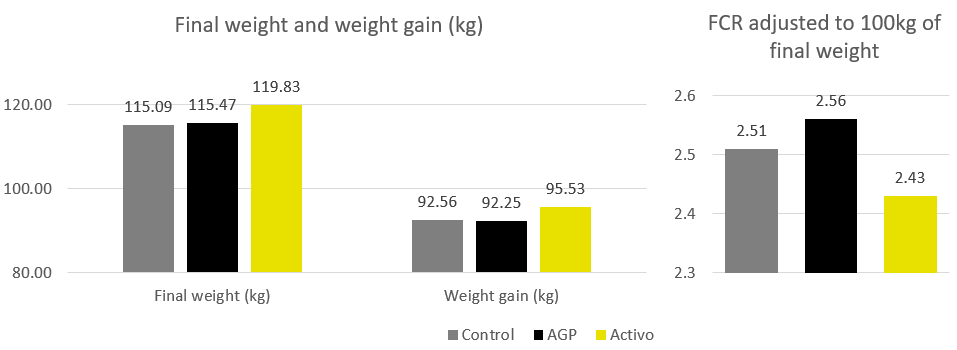
Concerning growth performance, the group fed the phytomolecules-based product Activo showed a 4.36 kg higher final weight after 126 days than the group provided AGPs (p=0.11), resulting in a 3.28 kg higher weight gain (p=0.21) and a 13 points better feed conversion.
Figure 2. Data of growth performance including final weight, weight gain and FCR adjusted to 100kg
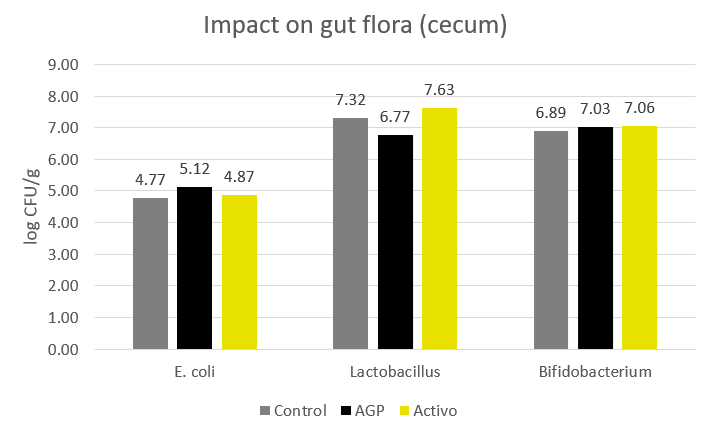
The evaluation of some bacteria naturally occurring in the gut flora showed that, in contrast to the antibiotic prophylaxes, Activo has no negative impact on E. coli, Lactobacillus and Bifidobacterium. However, the antibiotic group showed a slight decrease in the population of Lactobacilli (Figure 3).
Figure 3. Impact of antibiotics and phytomolecules (Activo) on the composition of the gut flora
This trial shows Activo increasing growth performance and feed conversion without any negative impact on gut flora. The addition of phytomolecules (Activo) to the feed is documented as optimal long-term support instead of antibiotic growth promoters.
Trial shows: Phytomolecules support animals in critical situations like weaning
In a trial conducted in the USA, a product containing phytomolecules and organic acids (Activo Liquid, EW Nutrition) was compared to an antibiotic for controlling bacterial diseases in US pig production (Mecadox). For the trial, a total of 360 weanling pigs, about 19 days old and weighing 5.70 kg, were divided into four groups. Each group consists of 9 pens with 10 animals per pen. All groups were fed a 3-phase diet.
To the different trial groups, the following products were added (table 2):
| Feeding valid for all groups | Group / Product | Inclusion rate and period of application | |
| 3-phase feeding after weaning: | Mecadox | 50 g/t of feed during the whole period | |
| Phase I (days 0-7): | 23 % CP, 5.4 % CF | Activo Liquid 3 | 375 ml/1000 L of water for 3 days post-weaning |
| Phase II (days 8-21): | 21 % CP, 4.1 % CF | Activo Liquid 5 | 375 ml/1000 L of water for 5 days post-weaning |
| Phase III (days 22-42): | 19 % CP, 4.4 % CF | Activo Liquid 7 | 375 ml/1000 L of water for 7 days post-weaning |
These performance parameters were evaluated: live weight, daily gain, daily feed intake, feed:gain ratios, and mortality.
Table 2. Feeding and inclusion of the additives
Results
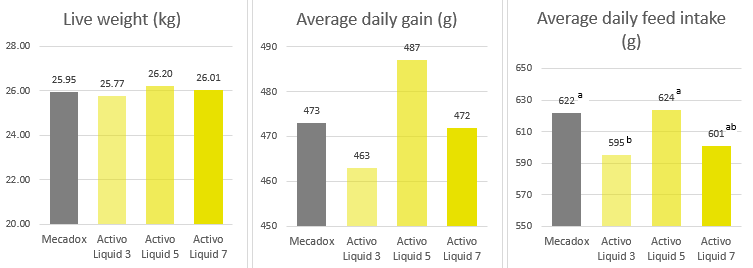
The results of the trial are shown in figure 4. Concerning growth, no significant differences could be seen between the groups, only numerical differences. Live weight in the antibiotic group was 25.95 kg, and in the Activo Liquid groups, it ranges from 25.77 kg (shortest period of application) to 26.20 kg (see below). This resulted in calculated values for an average daily gain of 473 g in the Mecadox fed animals and 463 to 487g in the Activo Liquid groups. Due to a lower feed intake per kg of weight gain, all groups fed Activo Liquid showed a significantly (p=0.05) better feed conversion than the Mecadox group.
Figure 4. Live weight in the groups fed the antibiotic Mecadox and the phytomolecules-based product Activo Liquid for different periods
Average daily gain in the different trial groups
Average daily feed intake in the different trial groups (P=0.05)
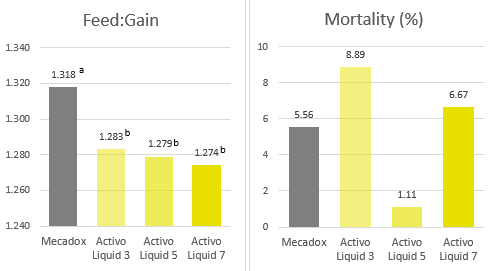
Concerning mortality, the group fed Activo Liquid for 5 days showed the lowest mortality rate of 1.1% (figure 5).
Figure 5. Feed:gain ratio in the different trial groups (P=0.05) & Mortality rates
Considering all parameters, the group fed Activo Liquid for five days provided the best results: numerically the lowest mortality rate, highest daily gain, and one of the two lowest feed:gain ratios. This trial concludes that Activo Liquid with an application period of five days can safely replace antibiotic growth promoters in the diet. Therefore, Activo Liquid is an interesting tool to additionally support pigs during critical periods of life.
Phytomolecules help keep health and performance together
The trials conducted with two types of phytomolecules-based products show that phytomolecules efficiently support pigs to achieve their genetic potential. A basic supply of these substances within the feed yields results similar to those of animals receiving antibiotic growth promoters (AGPs). In challenging situations like weaning, additional short-term supply is recommended, which can be done with liquid products via the waterline.
With this strategy, the use of antibiotic growth promoters and, therefore, antibiotics in general can be drastically reduced. This approach can help decrease antimicrobial resistance and, not to be forgotten, accommodates final customers’ requests for the lower usage of antibiotics in livestock.
References
Barber, Sarah, and Nikki Sutherland. “O’Neill Review into Antibiotic Resistance.” House of Commons Library, March 6, 2017. https://commonslibrary.parliament.uk/research-briefings/cdp-2017-0074/.
Baser, Kemal Hüsnü Can, and Gerhard Buchbauer. Handbook of Essential Oils: Science, Technology, and Applications. Boca Raton, FL: Taylor & Francis distributor, 2010.
Davies, Dame Sally. “Antibiotic Resistance ‘Big Threat to Health’.” BBC News. BBC, November 16, 2012. https://www.bbc.co.uk/news/health-20354536.
De Greeff, S.C., A.F. Schoffelen, and C.M. Verduin. “MARAN Reports.” WUR. National Institute for Public Health and the Environment – Ministery of Health, Welfare and Sport, June 2020. https://www.wur.nl/en/Research-Results/Research-Institutes/Bioveterinary-Research/In-the-spotlight/Antibiotic-resistance/MARAN-reports.htm.
Hao, Haihong, Guyue Cheng, Zahid Iqbal, Xiaohui Ai, Hafiz I. Hussain, Lingli Huang, Menghong Dai, Yulian Wang, Zhenli Liu, and Zonghui Yuan. “Benefits and Risks of Antimicrobial Use in Food-Producing Animals.” Frontiers in Microbiology 5, no. Art. 288 (2014): 1–11. https://doi.org/10.3389/fmicb.2014.00288.
Marquardt, Ronald R, and Suzhen Li. “Antimicrobial Resistance in Livestock: Advances and Alternatives to Antibiotics.” Animal Frontiers 8, no. 2 (2018): 30–37. https://doi.org/10.1093/af/vfy001.
O’Neill, J. “Tackling Drug-Resistant Infections Globally.” Review on Antimicrobial Resistance. Wellcome Trust / HM Government, May 19, 2016. https://amr-review.org/sites/default/files/160518_Final%20paper_with%20cover.pdf.