What oxidative stress and inflammation have to do with it, why it affects gut health, and how in-feed products support mitigation strategies
Stress in animals can be defined as any factor causing disruptions to their homeostasis, their stable internal balance. Stress engenders a biological response to regain equilibrium. High environmental temperatures are among the most important environmental stressors for poultry production, causing significant economic losses for the industry.
Climate change, thermoregulation, and stress
Climate change has increased the prevalence and intensity of heat stress conditions in most poultry production areas all over the world.
The optimum temperature for poultry animals’ well-being and performance –the so-called thermoneutral zone– is between 18 and 22°C. When birds are kept within this temperature range, they do not have to spend energy on maintaining constant body temperature.
Heat stress is the result of unsuccessful thermoregulation in the animals, as they produce a higher quantity of heat than they can lose. It means that there is a negative balance between the net amount of heat produced by the animal and its capacity to dissipate this body heat to the environment.
Heat stress – contributing factors
This energy imbalance is influenced by environmental factors such as sunlight, thermal irradiation, air temperature, humidity, and stocking density, but also by animal-related factors such as body weight, feather coverage and distribution, hydration status, metabolic rate, and thermoregulatory mechanisms. Moreover, stressors can be additive and different factors such as feed quality and disease can convene leading to severe losses in health and performance.
Increasing the respiratory rate -panting- is the main mechanism of chickens to loss heat, which is achieve by the evaporation of water from the respiratory tract however, relative humidity imposes a ceiling on water evaporation and subsequent dissipation of heat. Thus, the association of heat stress not only with high temperature, but also with high relative humidity.
Heat stress can be classified into two main categories, acute and chronic:
- Acute heat stress refers to a short and fast increase in environmental temperature (a few hours), in general, poultry animals show a degree of resilience to acute heat stress.
- Chronic heat stress is when the high temperatures persist for more extended periods (several days), and their compensatory mechanisms are not sufficient to maintain tissue integrity and thus health and performance are hindered.
The animal’s response to heat stress
When the environmental temperature is above the thermoneutral zone, the animals activate thermoregulation mechanisms to lose heat through behavioral, biochemical, and physiological changes and responses.
Behavioral changes
Panting and exposure of low/non-feathered body areas (raising wings) are the main behavioral mechanisms in which chickens regulate their body temperature when exposed to heat stress. These actions help the chickens to cool down, at a high toll: high energy demands, dehydration, respiratory alkalosis, lethargy, decrease in feed intake, loss of intestinal function and oxidative stress.
Physiological changes
The cardiovascular system also responds to high temperatures by deviating blood to the peripheral areas of the body to maximize the dissipation of heat. This implicates a reduced supply of nutrients and oxygen to the gastrointestinal tract, hindering its functions and provoking inflammation and oxidative stress.
The hypothalamic-pituitary-adrenal (HPA) axis gets activated, increasing the levels of circulating corticosterone, skeletal protein synthesis and the immune system is suppressed, therefore the animals stop growing and are more susceptible to disease.
Heat stress also changes the gene expression of cytokines, upregulates heat shock proteins (HSP), and reduces the concentration of thyroid hormones. When heat stress persists, these cascades of cellular reactions result in tissue damage and malfunction. The animals exposed to heat stress suffer adverse effects in terms of performance, which are widely known and include high mortality, lower growth, and production (Figure 1), and a decline in meat and egg quality.
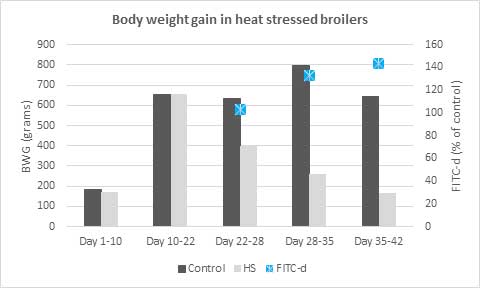 Figure 1: Body weight gain of broilers exposed to chronic heat stress (35°C continuously from day 21). A marker for tight junction permeability was added to feed (FITC-d – fluorescein isothiocyanate dextran); its fluorescence (in serum) increased with heat stress exposure time, showing higher intestinal permeability.
Figure 1: Body weight gain of broilers exposed to chronic heat stress (35°C continuously from day 21). A marker for tight junction permeability was added to feed (FITC-d – fluorescein isothiocyanate dextran); its fluorescence (in serum) increased with heat stress exposure time, showing higher intestinal permeability.
(Adapted from Ruff et al., 2020)
Outcomes of heat stress
Oxidative stress
Oxidative stress, simply put, occurs when the amount of reactive oxygen species (ROS) and nitrogen reactive species (NRS), exceed the antioxidant capacity of the cells. Oxidative stress is regarded as one of the most critical stressors in poultry production as it is a response to diverse challenges affecting the animals.
The normal metabolism of the animal – its energy production – generates ROS and RNS, such as hydroxyl radicals, superoxide anions, hydrogen peroxide, and nitric oxide. These usually are further processed by antioxidant enzymes produced by the cell, including superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px). Nutrients such as selenium and vitamins E, C, and A also participate in antioxidant processes. When the generation of ROS exceeds the capacity of the antioxidant system, oxidative stress ensues.
Heat stress leads to higher cellular energy demand, promoting an overload of ROS in the mitochondria. Consequently, oxidative stress occurs in several tissues, leading to cell apoptosis or necrosis as oxidized molecules can take electrons from other molecules, resulting in a chain reaction. Among these tissues, the gastrointestinal tract can be highly affected.
Impaired gut function
In the gastrointestinal tract, oxidative stress and the consequent tissue damage, lower feed digestion and absorption, increase intestinal permeability and modify the microbiome.
Changes in intestinal morphology and digestive function
Heat stress affects intestinal weight, length, barrier function, and microbiota, resulting in animals that have lower total and relative weight of the small intestine, with shorter jejunum and duodenum, shorter villi (Figure 2), and reduced absorption areas, in comparison to non-stressed animals.
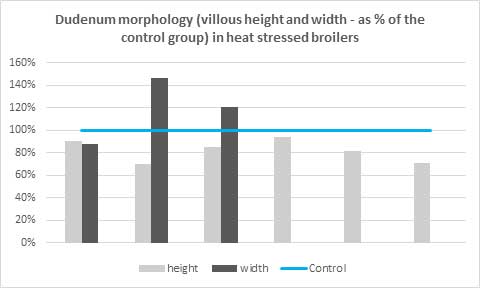 Figure 2: Villous height and width of broilers exposed to heat stress in relation with the control group (100%). Villous height is always shorter than the control group, but width can increase as the organisms shows resilience to the stressful situations and aims to recover intestinal surface. (Adapted from Jahejo et al., 2016; Santos et al., 2019; Wu et al., 2018; Abdelqader et al., 2016 ; Santos et al., 2015 and Awad et al., 2018 – by order of appearance in the graph from left to right)
Figure 2: Villous height and width of broilers exposed to heat stress in relation with the control group (100%). Villous height is always shorter than the control group, but width can increase as the organisms shows resilience to the stressful situations and aims to recover intestinal surface. (Adapted from Jahejo et al., 2016; Santos et al., 2019; Wu et al., 2018; Abdelqader et al., 2016 ; Santos et al., 2015 and Awad et al., 2018 – by order of appearance in the graph from left to right)
Changes in the intestinal microbiome
Due to reduced feed intake and impaired intestinal function, the presence and activity of the commensal microbiota can also be modified. Heat stress can lead to reduced populations of beneficial microbes, boost the growth of potential pathogens leading to dysbiosis and necrotic enteritis.
Changes in intestinal permeability
Several studies indicate that both acute and chronic heat stress increase gut permeability, not only by lowering feed intake, but also by increasing intestinal oxidative stress and disrupting the expression of tight junction proteins.
Heat and oxidative stress in the gut result in cell injury and apoptosis. When the tight junction barrier is compromised, luminal substances leak into the bloodstream, which constitutes the condition known as “leaky gut”. This includes the translocation of pathogenic bacteria, including zoonotic pathogens (e.g. Salmonella and Campylobacter); consequently, a higher risk of contamination of food products can be expected.
Endotoxins
Bacterial lipopolysaccharides (LPS), also known as endotoxins, constitute the main components of the outer membrane of all gram-negative bacteria and are essential for their survival. LPS have direct contact with the bacteria’s surroundings. They function as a protection mechanism against the host’s immunological response and chemical attacks from bile salts, lysozymes, or other antimicrobial agents.
Gram-negative bacteria are part of poultry animals’ microbiota; thus, there are always LPS in the intestine. Under optimal conditions, this does not affect animals because intestinal epithelial cells are not responsive to LPS when stimulated from the apical side. In stress situations, the intestinal barrier function is impaired, allowing the passage of endotoxins into the blood stream. When LPS are detected by the immune system either in the blood or in the basolateral side of the intestine, inflammation and changes in the gut epithelial structure and functionality occur.
An increased release and passage of endotoxins has been demonstrated in heat stress (Figure 3) as well as a higher expression of TLR-4 and other inflammation biomarkers, which contributes to the deleterious effects of heat stress in the animals. Moreover, blood LPS induces systemic inflammatory reactions that force the organism to divert energy to support the immune system which furthermore depresses performance.
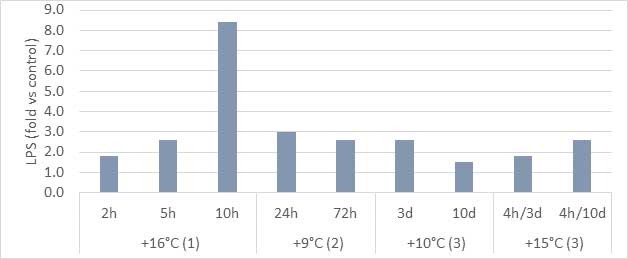 Figure 3 – Systemic LPS increase (in comparison with a non-stressed control) after different heat stress challenges in broilers:16°C increased for 2, 5 and 10 hours (Huang et al., 2018); 9°C increased for 24 and 72 hours (Nanto-Hara et al., 2020); 10°C continuously for 3 and 10 days, and 15°C 4 hours daily for 3 and 10 days (Alhenaky et al., 2017).
Figure 3 – Systemic LPS increase (in comparison with a non-stressed control) after different heat stress challenges in broilers:16°C increased for 2, 5 and 10 hours (Huang et al., 2018); 9°C increased for 24 and 72 hours (Nanto-Hara et al., 2020); 10°C continuously for 3 and 10 days, and 15°C 4 hours daily for 3 and 10 days (Alhenaky et al., 2017).
Mitigation strategies
Most intervention strategies deal with heat stress through a wide range of measures, including environmental management, housing design, ventilation, sprinkling, and shading, amongst others. Understanding and controlling environmental conditions is a crucial part of heat stress management.
Feed management and nutrition interventions are also recommended to reduce the effects of heat stress. They include feeding pelletized diets with increased energy coming from fats and oils, reduction of total protein with additional supplemental amino acids, increasing levels of vitamins and minerals, and adjusting the dietary electrolyte balance.
Antioxidants
Under oxidative stress conditions in the gut, there is a demand for antioxidants to counteract the excess of ROS; hence, dietary antioxidants can help reduce ROS and improve animal performance.
Research shows that certain phytomolecules, including thymol, carvacrol, cinnamaldehyde, silybinin and quercetin have antioxidant properties and improve performance under conditions of oxidative stress. The antioxidant capacity of phytomolecules manifests itself in free radical scavenging, increased production of natural antioxidants, and the activation of transcription factors. Moreover, menthol and cineol, also aid animals under heat stress by simulating the sensory cold receptors of the oral mucosa, giving the animals a cooling sensation, and reducing heat stress behavior.
Controlling LPS and oxidative stress
An experiment conducted by EW Nutrition GmbH had the objective to evaluate the ability of a product (Solis Max 2.0) in mitigating heat-stress induced LPS as well as oxidative stress.
For the experiment, Cobb 500 breeder pullets were divided in two groups, each group was placed in 11 pens of 80 hens, in a single house. One of the groups received feed containing 2kg/ton of the product from the first day. From week 8 to week 12, the temperature of the house was raised 10°C for 8 hours every day.
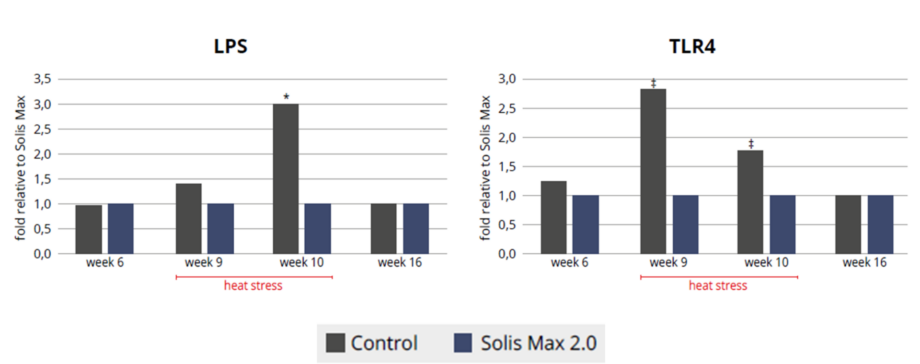
Figure 4 and 5: Blood LPS and expression of toll-like receptor 4 (TLR4) in lymphocites of pullets before (wk 6), and during heat stress (wk 9 and 10). (*) indicates significant differences (P<0,05), and (‡) a tendency to be different against the control group (P<0,1).
Throughout the heat stress period, blood LPS (Fig 4) was lower in the pullets receiving the product, which allowed lower inflammation evidenced by the lower expression of TLR4 (Fig. 5). Oxidative stress was also mitigated with the help of the combination of phytomolecules in the product (Fig. 6), obtaining 8.5% improvement on serum total antioxidant capacity (TAC), supported by an increase in in superoxide dismutase (SOD glutathione peroxidase (GSH) and a decrease in malondialdehyde (MDH).
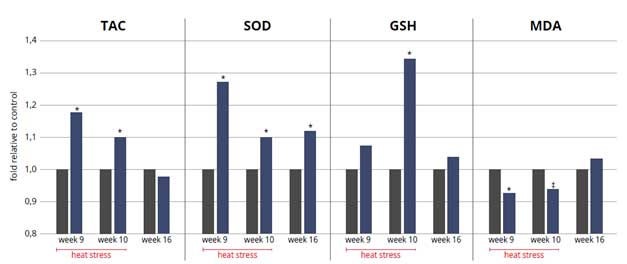 Figure 6: Antioxidant capacity of pullets during heat stress (wk 9 and 10). (*) indicates significant differences (P<0,05), and (‡) a tendency to be different against the control group (P<0,1). Parameters measured are total antioxidant capacity (TAC), super oxide dismutase (SOD), gluthatione peroxidase (GSH), and malondialdehyde (MDA).
Figure 6: Antioxidant capacity of pullets during heat stress (wk 9 and 10). (*) indicates significant differences (P<0,05), and (‡) a tendency to be different against the control group (P<0,1). Parameters measured are total antioxidant capacity (TAC), super oxide dismutase (SOD), gluthatione peroxidase (GSH), and malondialdehyde (MDA).
In the bottom line, the heat stress challenge also affected performance, affecting feed conversion (9 points lower) and body weight (3% lower). The optimal supporting product was able to efficiently reduce the LPS exposure for the pullets and thus inflammation and oxidative stress were reduced, as a consequence energy could be driven to performance evidenced by a better BW and FCR.
Summary
Heat stress is a common reality in poultry production, its effects are quite complex and harmful and depend on the intensity and duration of the exposure to high temperatures.
By lowering feed digestibility, increasing gut permeability, and compromising immunity, heat stress leaves animals more susceptible to gut-health related issues such as dysbacteriosis and necrotic enteritis – and thus may increase the need to use antibiotics. Additionally, the passage of LPS through the permeable gut induces inflammation and further damage to animal welfare, health and performance.
Mitigation strategies, including support to the gut oxidative balance and lowering LPS-induced inflammation are crucial to support poultry animals in these critical periods.
References:
- Das, S. et al., 2011. Nutrition in relation to diseases and heat stress in poultry. Veterinary World, 4(9), pp. 429-432.
- Surai, P. F., Kochish, I. I., Fisinin, V. I. & Kidd, M. T., 2019. Antioxidant defense systems and oxidative stress in poultry biology: An update. Antioxidants, 8(7).
- St-Pierre, N., Cobanov, B. & Schnitkey, G., 2003. Economic Losses from Heat Stress by US Livestock Industries. Journal of Dairy Science, Volume 86
- Tellez Jr., G., Tellez-Isaias, G. & Dridi, S., 2017. Heat stress and gut health in broilers: role of tight junction proteins. Advances in Food Technology and Nutritional Sciences, 3(1).
- Lian, P. et al., 2020. Beyond heat stress: intestinal integrity disruption and mechanism-based intervention strategies. Nutrients, Volume 12.
- Akbarian, A. et al., 2016. Association between heat stress and oxidative stress in poultry; mitochondrial dysfunction and dietary interventions with phytochemicals. Journal of Animal Science and Biotechnology, 7(37).
- Lara, L. & Rostagno, M., 2013. Impact of heat stress on poultry production. Animals, Volume 3, pp. 356-369.
- Saeed, M. et al., 2019. Heat stress management in poultry farms: a comprehensive overview. Journal of Thermal Biology, Volume 84, pp. 414-425.
- Farag, M. & Alagawany, M., 2018. Physiological alterations of poultry to the high environmental temperature. Journal of Thermal Biology, Volume 76, pp. 101-106.
- Quinteiro-Filho, W. et al., 2010. Heat stress impairs performance parameters, induces intestinal injury, and decreases macrophage activity in broiler chickens. Poultry Science, Volume 89, p. 1905–1914.
- Santos, R. et al., 2015. Quantitative histomorphometric analysis of heat-stress-related damage in the small intestines of broiler chickens. Avian Pathology, 44(1), pp. 19-22.
- Awad, E. et al., 2018. Growth performance, duodenal morphology, and the caecal microbial population in female broiler chickens fed glycine-fortified low protein diets under heat stress conditions. British Poultry Science, 59(3), pp. 340-348.
- Mujahid, A., Yoshiki, Y., Akiba, Y. & Toyomizu, M., 2005. Superoxide radical production in chicken skeletal muscle induced by heat stress. Volume 84, pp. 307-314.
- Hu, R. et al., 2019. Polyphenols as potential attenuators of heat stress in poultry production. Antioxidants, 8(67).
- Salami, S. et al., 2015. Efficacy of dietary antioxidants on broiler oxidative stress, performance and meat quality: science and market. Avian Biology Research, 8(2), pp. 65-78.
- Lauridsen, C., 2019. From oxidative stress to inflammation: redox balance and immune system. Poultry Science, Volume 98, pp. 4240-4246.
- Surai, P. F. & Fisinin, V. I., 2016. Vitagenes in poultry production: Part 1. Technological and environmental stresses. World’s Poultry Science Journal, Volume 72.
- Arab Ameri, S., Samadi, F., Dastar, B. & Zarehdaran, S., 2016. Efficiency of peppermint (Mentha piperita) powder on performance, body temperature, and carcass characteristics of broiler chickens in heat stress condition. Iranian Journal of Applied Animal Science, 6(4), pp. 943-950.
- Saadat Shad, H., Mazhari, M., Esmaeilipour, O. & Khosravinia, H., 2016. Effects of thymol and carvacrol on productive performance, antioxidant enzyme activity, and certain blood metabolites in heat-stressed broilers. Iranian Journal of Applied Animal Science, 6(1), pp. 195-202.
- Mishra, B. & Jha, R., 2019. Oxidative stress in the poultry gut: potential challenge and interventions. Frontiers in Veterinary Science, 6(60).
- Ruff, J. et al., 2020. Research Note: Evaluation of a heat stress model to induce gastrointestinal leakage in broiler chickens. Poultry Science, Volume 99, pp. 1687-1692.
- Rostagno, M., 2020. Effects of heat stress on the gut health of poultry. Journal of Animal Science, 98(4).
- Abdelqader, A. & Al-Fataftah, A., 2016. Effect of dietary butyric acid on performance, intestinal morphology, microflora composition and intestinal recovery of heat-stressed broilers. Livestock Science, Volume 183.
- Jahejo, A. et al., 2016. Effect of heat stress and ascorbic acid on gut morphology of broiler chicken. Sindh University Research Journal, 48(4), pp. 829-832.
- Wu, Q. et al., 2018. Glutamine alleviates heat stress-induced impairment of intestinal morphology, intestinal inflammatory response, and barrier integrity in broilers. Poultry Science, Volume 97, pp. 2675-2683.
- Santos, R. et al., 2019. Effects of a feed additive blend on broilers challenged with heat stress. Avian Pathology, 48(6), pp. 582-601.
- Shi, D. et al., 2019. Impact of gut microbiota structure in heat-stressed broilers. Poultry Science, Volume 98, pp. 2405-2413.
- Burkholder, K. et al., 2008. Influence of stressors on normal intestinal microbiota, intestinal morphology, and susceptibility to Salmonella Enteritidis colonization in broilers. Poultry Science, Volume 87, pp. 1734-1741.
- Quinteiro-Filho, W. et al., 2012. Acute heat stress impairs performance parameters and induces mild intestinal enteritis in broiler chickens: the role of acute HPA axis activation. Journal of Animal Science.
- Antonissen, G. et al., 2014. The Impact of Fusarium Mycotoxins on Human and Animal Host Susceptibility to Infectious Diseases. Toxins, 6(2).
- Tsiouris, V. et al., 2018. Heat stress as a predisposing factor for necrotic enteritis in broiler chicks. Avian Pathology, 47(6), pp. 616-624.
- Abd El-Hack, M. et al., 2019. Herbs as thermoregulatory agents in poultry: An overview. Science of the Total Environment.
- Surai, P. F., 2020. Antioxidants in poultry nutrition and reproduction: An update. Antioxidants, 9(2).
- Surai, P. F., 2015. Silymarin as a natural antioxidant: An overview of the current evidence and perspectives. Antioxidants, 4(1).
- El-Maaty, A., Hayam, M., Rabie, M. & El-Khateeb, A., 2014. Response of heat-stressed broiler chicks to dietary supplementation with some commercial herbs. Asian Journal of Animal and Veterinary Advances, 9(12), pp. 743-755.
- Prieto, M. & Campo, J., 2010. Effect of heat and several additives related to stress levels on fluctuating asymmetry, heterophil:lymphocyte ratio, and tonic immobility duration in White Leghorn chicks. Poultry Science, Volume 89, p. 2071–2077.
- Beckford R. C., Ellestad L. E., Proszkowiec-Weglarz M., Farley L., Brady K., Angel R., et al. 2020. Effects of heat stress on performance, blood chemistry, and hypothalamic and pituitary mRNA expression in broiler chickens. Poult. Sci. 99, 6317–6325.
- Brugaletta G., Teyssier J. R., Rochell S. J., Dridi S., Sirri F. 2022. A review of heat stress in chickens. Part I: Insight into gut health and physiology. Front. Physiol. Avian Physiology. Volume 13 – 2022