What happens to everything else if the food system fails? … And why we should make sure that doesn’t happen

by Ilinca Anghelescu, Global Director Marketing & Communications, EW Nutrition
Every week, a new story promises to change how we eat. Lab-grown steaks. Vertical farms fed by LED lights. Cricket flour. The algae revolution. Regenerative everything.
Meanwhile, somewhere in Iowa, a farmer is managing soil drainage at 4 a.m. In the Yangtze River Delta, flooded paddy fields are being leveled by laser-guided equipment. In the Sahel, sorghum is being harvested by hand under brutal heat. In the Netherlands, greenhouse engineers are coaxing eight tomato harvests a year from hydroponic systems. Such professionals, such practices are, collectively, the reason 8 billion people ate today.
How we got here, and why we cannot go back
The density problem nobody talks about
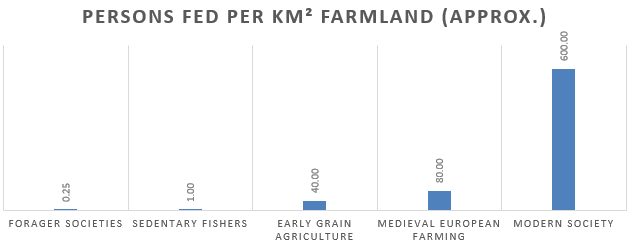
In his 2024 book How to Feed the World, Czech-Canadian professor and researcher Vaclav Smil notes that, across 300 forager societies that persisted into the 19th and 20th centuries, the mean population density was 0.25 persons per square kilometer.1 The most productive forager groups, those with access to salmon runs or seal hunting on Pacific coastlines, could reach just above one person per square kilometer. By contrast, intensive agricultural systems in southern China during the Qing dynasty supported more than 500 people per square kilometer of farmland.1 Contemporary industrial agriculture can support between 500-900.
In Smil’s analysis, agriculture is not slightly more efficient at feeding people than foraging. Agriculture is between 500 and 2,000 times more efficient than foraging.
The thought experiment Smil runs through disposes of several popular fantasies at once, including those in which humans go back to a primitive way of eating. For instance, an adult human eating like a chimpanzee (roughly 80 percent fruit by mass) would need four to five kilograms of ripe fruit daily, requiring hours of foraging and providing almost no fat or protein.1 To supply just the European Union’s 450 million people with adequate protein via this dietary route would require more than half a billion tons of figs per year, roughly 400 times the entire 2020 global fig harvest.1 The chimp model, like other primitive models (whether purely foraging or hunting or a mixed model), cannot scale.
In other words, in a world currently trying to feed 8.3 billion people, the transition to agriculture cannot be undone.
The rule of 20: Why we eat so few plants
One of the more counterintuitive facts in food systems science is how narrow our dietary base actually is. Botanists have classified nearly 400,000 species of vascular plants. Roughly 12,000 of those are grasses capable of producing nutritious seeds. Of these, humanity has domesticated a tiny fraction. Just 20 plant species account for 75 percent of all annually harvested crops by weight. Two of those species, rice and wheat, alone supply 35 percent of global food energy.1
This is not a failure of agricultural imagination but the result of stringent selection criteria that operated over thousands of years. Smil calls these criteria the “entry requirements” for staple crops: fast maturation, high yield, long shelf life, resistance to pests, and high energy density. Wheat, for example, contains roughly 350 kilocalories per 100 grams. Tomatoes contain fewer than 20 kcal/100g. Wheat is 18 times more energy-dense per unit weight.1
The early civilizations that independently discovered the cereal-legume combination (corn and beans in the Americas, rice and soybeans in Asia, wheat and lentils in the Middle East) were solving an amino acid optimization problem without knowing it. Cereals are low in the essential amino acid lysine. Legumes are high in it. Together, they provide a complete protein profile. The world’s great cuisines, from Mexican rice and beans to Japanese miso soup over rice, are not accidents. They are dietary solutions that natural selection, mediated through human survival and culture, arrived at over millennia.1

What the economy doesn’t count
The GDP illusion
In standard economic accounting, agriculture contributes roughly 1 to 4 percent of GDP in developed countries and somewhat more in developing ones. This number is cited constantly as evidence that farming is a residual sector, economically marginal, safely neglected in favor of “shinier” industries.
Smil dismantles this framing methodically. When you add food processing, food manufacturing, beverages, food retail, and food service, the food system in the United States accounts for approximately 5 percent of GDP and more than 10 percent of total employment.1 But even this number, broad as it is, underestimates the true scale, because it fails to capture the full infrastructure dependency: the fuel and energy consumed by agricultural machinery, the chemical industry built to supply fertilizer, the logistics networks dedicated to food transport and cold chain management, and the healthcare costs tied to diet-related disease.
When Smil attempts a full-system accounting of global food, including production, processing, transportation, wholesale, retail, storage, and consumption, he concludes that the food system’s true share of global economic activity is on the order of 25 to 30 percent of respective totals, with standard economic accounts attributing less than 5 percent representing “grossly inaccurate and highly misleading quantifications.”1
The energy picture is similarly startling. Smil calculates that the global food system consumes between 20 and 25 percent of the world’s annual primary energy supply.1 This includes the energy to grow, harvest, process, refrigerate, transport, package, cook, and dispose of food. It is the single largest category of energy use in human civilization, larger than personal transportation, larger than industrial manufacturing of most goods, and yet it rarely appears in climate policy discussions with the prominence its scale demands.
Smil offers one striking comparison that has only sharpened since his original analysis. The global smartphone market in 2024 generated approximately $441 billion in wholesale revenue, calculated from approximately 1.24 billion units shipped at a record average selling price of $356.34 In that same year, the global wheat harvest, some 799 million tons, was worth approximately $215 billion at reference export prices, and the global rice harvest of roughly 541 million tons was worth approximately $318 billion.32 33 Combined, just these two crops generated an estimated $533 billion, roughly 20 percent more than the entire global smartphone market. Two crops, grown on a fraction of Earth’s farmland, produced economic value that exceeds the most ubiquitous consumer technology device in human history.

Revolutions usually come from empty stomachs
A history lesson worth remembering
The historical relationship between food insecurity and political instability is one of the most robustly documented relationships in social science. The French Revolution of 1789 was preceded by catastrophic grain harvests in 1788. Bread prices in Paris in early 1789 consumed up to 88 percent of a worker’s daily wage.2 The Arab Spring of 2010-2011 was triggered, at least in part, by a spike in global food commodity prices. Mohamed Bouazizi, the Tunisian street vendor whose self-immolation catalyzed a regional uprising, was a food vendor who had his produce confiscated.3
The research is consistent. A 2011 preprint study published by Marco Lagi and colleagues at the New England Complex Systems Institute found that global food price spikes, as measured by the FAO Food Price Index, were a consistent precursor to social unrest and political instability events across multiple continents.3 A 2015 paper in the American Journal of Agricultural Economics extended this analysis, finding statistically significant relationships between cereal price levels and social unrest.4
The baseline condition for social order is that people have access to food. Everything else, including the liberal democratic institutions, the tech economies, and the climate negotiations that dominate contemporary policy attention, depends on that foundation being intact. Smil makes this point in structural rather than historical terms. When he asks whether smartphones or food matter more, the answer is obvious to him: “A world without smartphones would be poorer and less convenient. A world without food would not exist.”1
The 9%
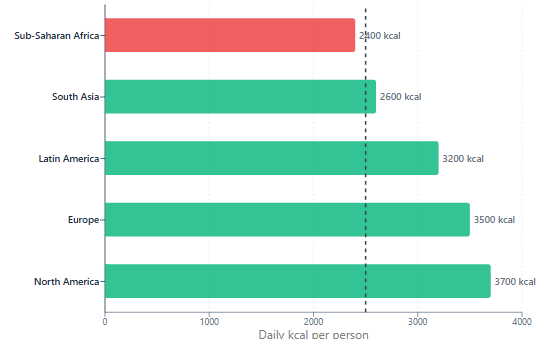
According to the UN Food and Agriculture Organization, approximately 733 million people, roughly 9 percent of the global population, were undernourished in 2023.5 This is not primarily a production problem. As Smil notes and the FAO confirms, global food production averages around 3,000 kilocalories per person per day, which is substantially above the roughly 2,500 kilocalories required by an average active adult.15 The world produces enough calories to feed everyone.
The problem is access, poverty, and distribution. Hunger is a political economy failure, as price spikes hit the poor first and hardest. But if global food production fell by 10 percent, the 9 percent who are currently undernourished would not be the only ones suffering. Supply shocks ripple through markets and a globalized world does not allow for compartmentalized impact as much as it used to.
The real environmental cost: Agriculture and alternatives
Some immediate problems have immediate solutions
Agriculture accounts for approximately 72 percent of global freshwater withdrawals.1 Cropland and permanent pastures together cover about 36 percent of non-glaciated land.1 The food system is responsible for approximately 34 percent of global greenhouse gas emissions, based on the most comprehensive analysis available.6 These figures are often presented as indictments. They should instead be understood as measures of necessity. The question is not “why does food production use so much?” but “what would we use it on instead, and would that work?”
The FAO’s global assessment of livestock’s climate impact, the famous 2006 report Livestock’s Long Shadow, attributed 18 percent of greenhouse gas emissions to livestock. A revised methodology in 2013, applying the same accounting framework used for other sectors, reduced this figure to approximately 14.5 percent.7
The nitrogen story is more nuanced. Smil notes that global nitrogen use efficiency (the share of applied fertilizer that ends up in harvested crop rather than escaping to air or water) averages around 40 percent globally, and has been falling in intensively farmed regions.1 In China, over-fertilization has driven efficiency from 37 percent down to 29 percent, with the difference escaping as nitrous oxide (a potent greenhouse gas), ammonia (an air pollutant), and nitrates (which contaminate groundwater and create coastal dead zones).1 This is a genuine problem with practical and affordable solutions: better timing of fertilizer application, matching fertilizer type to soil need, and precision agriculture technologies that reduce over-application.
The problems of industrial agriculture are, to a large extent, engineering problems. They have technical solutions that can be implemented incrementally, at scale, within existing agricultural systems. They do not require abandoning food production as we know it; they require improving it.
What “organic” actually means at scale
The appeal of organic farming as an environmental solution is real but its limits are underappreciated. A 2012 meta-analysis in Nature by Seufert and colleagues found that organic farming produces, on average, 25 percent lower yields than conventional farming across all crops, with the gap widening to 43 percent below conventional yields for some cereal crops.x8 A subsequent 2017 analysis in Agronomy for Sustainable Development by Lesur-Dumoulin and colleagues examining more than 50 studies found yield gaps of 19 to 25 percent, with significant variation by crop and region.x9
The implication is straightforward. Feeding the current global population on fully organic agriculture would require converting an additional 16 to 30 percent of the world’s remaining non-agricultural land to farmland, in order to compensate for lower yields.x10 The biodiversity loss from that land conversion would likely exceed the biodiversity gains from reduced pesticide use on existing farmland. This does not make organic farming in any way bad, it simply makes it a context-specific tool instead of a global solution.
Smil notes that in the centuries before synthetic fertilizers, when all farming was “organic” by definition, 80 percent of people worked in farming, doing physically exhausting work for marginal returns. The “liberation” of the majority of humanity from agricultural labor, one of the most profound quality-of-life improvements in history, was made possible by the Haber-Bosch process, the synthesis of ammonia from atmospheric nitrogen, invented in 1913. Without synthetic nitrogen fertilizer, global crop yields would fall by roughly 40 to 50 percent, and roughly half of the current human population could not be fed on existing farmland.x11
The alternatives don’t add up
Cultured meat: Promising, not a solution
The first cultured beef burger was produced in 2013 in the Netherlands at an estimated cost of $330,000.1 By 2020, Singapore approved the first commercial sale of cultured chicken nuggets, produced by Eat Just, at a price point still far above commodity chicken. By 2021, total investment in the sector had reached approximately $2 billion.1
The fundamental challenge is not biological but a matter of thermodynamics. Cultured meat production requires maintaining cells in a growth medium at controlled temperature and pH, with continuous oxygen supply, nutrient input, and waste removal. A 2023 preprint study by Risner and colleagues at UC Davis found that, under current production processes, the lifecycle greenhouse gas emissions of cultured beef could actually be higher than conventional beef over a 1,000-year time horizon, because the production of growth media requires large amounts of purified water and energy-intensive pharmaceutical-grade inputs.x12
The energy demand is particularly problematic. A 2019 analysis in Frontiers in Sustainable Food Systems by Lynch and Pierrehumbert (Oxford) found that cultured meat’s climate advantage over cattle depends heavily on whether energy production is decarbonized. Because cultured meat emissions are almost entirely CO₂ (which accumulates indefinitely) rather than methane, which breaks down within a decade, the long-term warming impact of cultured meat can exceed that of cattle under scenarios of continued high consumption. The energy advantage of cultured meat over monogastrics (pigs and poultry) is marginal at best and may reverse under realistic production conditions.”13
None of this means cultured meat has no future. It may eventually serve specific markets, particularly as a supplement to conventional production in regions where land is extremely constrained. But Smil’s verdict is clear: it is currently “pilot scale” technology, commercially unproven at mass market pricing, and it cannot meaningfully contribute to feeding up to 10 billion people in the next two to three decades.1
The vegan transition?
Beef is by far the largest emitter of CO2 equivalent per kilogram of protein, compared to chicken or pork.14 A diet shift from beef to other proteins in high-income countries would measurably reduce the food system’s climate impact.
But Smil flags an important caveat that often goes unmentioned in advocacy for plant-based diets: mass adoption of veganism in wealthy countries, if it leads to increased consumption of out-of-season fruits, nuts, avocados, and specialty protein crops, may not reduce and could even increase total environmental pressure.1 Almonds require approximately 12 liters of water per nut.15 Avocados, with their supply chains running from Mexico to Europe, have water footprints of approximately 320 liters per fruit and contribute to deforestation in growing regions.16
There is also a structural argument that rarely gets made: production animals serve functions beyond meat (and not even mentioning milk or eggs). Approximately 57 percent of current global livestock feed consists of materials that are not edible by humans: crop residues, grass from land unsuitable for cropping, and food processing byproducts such as oilseed cakes, bran, and distillers’ grains.17 Animals convert non-human-edible biomass into high-quality protein and fat. This is not waste but efficiency.
What Would Actually Work
First target waste
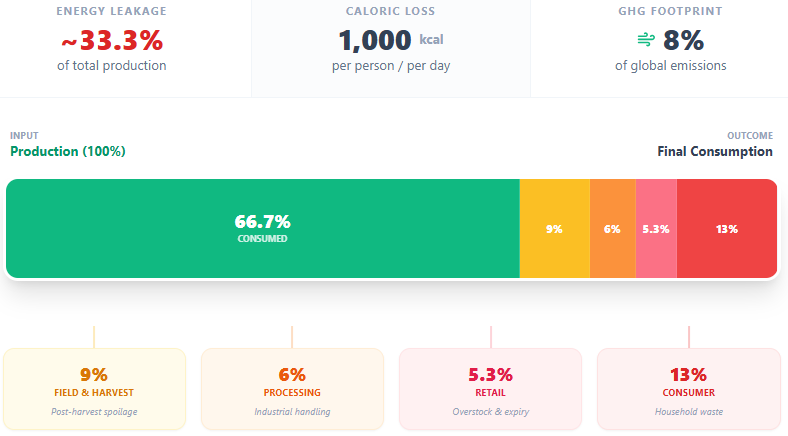
Global food waste amounts to approximately 1,000 kilocalories per person per day, roughly one-third of total food production.
The FAO estimates that approximately one-third of all food produced for human consumption, roughly 1.3 billion tons per year, is lost or wasted annually.18 Losses occur throughout the supply chain, from post-harvest spoilage in developing countries (where cold chain infrastructure is inadequate) to consumer behavior and retail overproduction in wealthy ones. The environmental cost of this waste is itself enormous: the production of food that is ultimately not eaten accounts for approximately 8 percent of global greenhouse gas emissions.19
The N fix that is already possible
Improving global nitrogen use efficiency (NUE) from its current 40 percent average to 60 to 65 percent, a target achievable through existing precision agriculture technologies (as mentioned before), would reduce the amount of synthetic nitrogen required to produce the current food output by roughly a third.20 This single change would decrease nitrous oxide emissions (which are 273 times more potent than CO2 over a 100-year timescale as a greenhouse gas, according to AR6, 2021 28), reduce freshwater nitrate contamination, and shrink coastal dead zones.
The technologies required are not exotic. Split nitrogen application (applying fertilizer in multiple smaller doses timed to crop uptake rather than one large dose at planting) can increase NUE by 15 to 20 percent with no change in yield.21 Soil testing and variable rate application technology, where GPS-guided equipment applies different fertilizer rates across a field based on measured soil nutrient levels, can improve NUE by a further 10 to 15 percent.22 These are available now, at commercially viable cost, for large-scale farming operations.
The barrier is not technical but rather economic and behavioral: fertilizer is cheap relative to its yield benefit, so farmers have limited financial incentive to apply it precisely. Policy tools, whether taxes on nitrogen over-application, payments for NUE improvements, or tighter limits on fertilizer application near waterways, could close this gap.
Meat mix and moderation
Smil estimates that approximately one-third of global cereal production and two-thirds of the US grain harvest are currently fed to animals.1 Feedlot beef carries a feed conversion ratio of roughly 30 kilograms of feed per kilogram of edible product at the high end.1 Poultry and pork convert feed to protein far more efficiently, and pasture-raised ruminants on land unsuitable for cropping represent a different calculation entirely.
The case for moderating high-end beef consumption in wealthy countries rests primarily on efficiency and emissions, not on the nutritional dispensability of meat as a food category. Meat, including beef, is a nutritionally dense and difficult-to-replicate protein source. It provides all essential amino acids in highly bioavailable form, along with heme iron, which is absorbed at rates of 15 to 35 percent compared to 2 to 20 percent for non-heme iron from plant sources, as well as zinc, vitamin B12, selenium, and conditionally essential compounds such as creatine and carnitine that are absent or negligible in unfortified plant foods.29 For populations in low- and middle-income countries where protein deficiency, iron deficiency, and micronutrient gaps remain widespread public health problems, the argument for reducing meat consumption requires a different cost-benefit analysis than it does in the United States or Northern Europe, where the concern is overconsumption rather than inadequacy.
The appropriate policy lever for high-income countries is therefore not elimination of meat categories but a shift in the composition of meat consumption toward more efficient and lower-emissions sources (more poultry and pork, less feedlot beef) while maintaining total protein adequacy. This is consistent with both the environmental evidence and updated dietary guidelines in major consuming nations. A 2016 analysis by Springmann and colleagues at Oxford, published in PNAS, found that transitioning toward diets in line with standard dietary guidelines could reduce global mortality by 6 to 10 percent and food-related greenhouse gas emissions by 29 to 70 percent compared with a 2050 reference scenario. 30 A subsequent 2018 modelling study by the same group in Nature confirmed that the dietary-guidelines scenario alone (without requiring full elimination of animal products) achieves a 29 percent reduction in food-related GHG emissions relative to projected baseline consumption.23 The gains are concentrated in high-income countries, and the modelling explicitly notes that applying the same dietary shift logic to low-income countries would in several cases increase land and water use rather than reduce it.31
Smil’s preferred framing holds: the goal is meat moderation and mix optimization, not categorical elimination.
What happens to everything else if the food system fails?
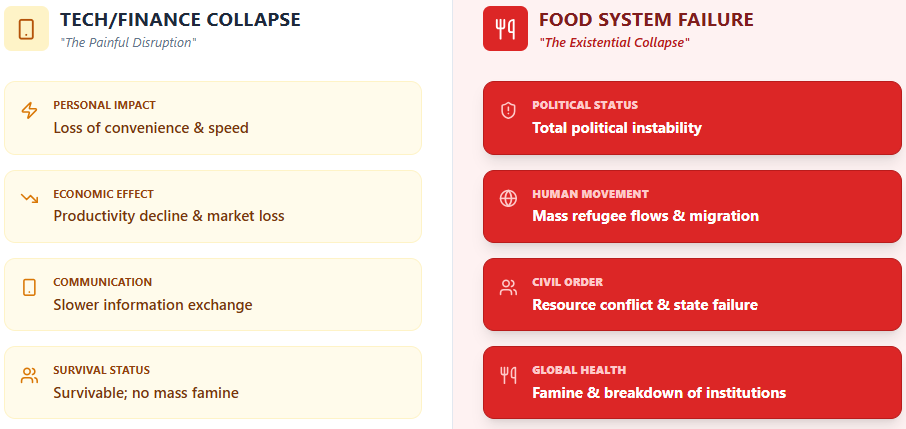
The answer is: everything collapses. Food insecurity at scale produces predictable cascades: political instability, refugee flows, conflict over resources, public health crises, and the breakdown of governance institutions that depend on social legitimacy. The Arab Spring, which reshaped the politics of a continent (and arguably the world), was triggered in part by a global food price spike following the 2010 Russian wheat export ban and droughts in major grain-producing regions.3
By contrast, the collapse of the smartphone market, while economically painful, would likely not produce famine, mass migration, or state failure. The collapse of social media platforms, though consequential for public discourse, would not endanger human life. The collapse of the global financial system, as catastrophic as the 2008 crisis demonstrated it could be, is survivable in ways that the collapse of food production is not.
The world needs to feed 9.7 billion people in 2050, according to the UN medium-population projection.24 The cultured meat industry cannot scale to meaningful market share within that timeframe under any realistic projection. Precision nitrogen management can, and is already beginning to, because it requires only incremental adoption of existing technology by existing farmers working existing land.
The nutritional transition that high-income countries have largely completed, from adequate calories to excess calories to dietary choice, is not yet available to much of the world’s population. Agricultural development policy that ignores this gradient would impose wealthy-world concerns on people or categories for whom adequate nutrition remains an unsolved problem.
Sustainability discourses must get priorities right
Food production is the prerequisite for everything else. Applying regulatory pressure to it without carefully calibrating the effects on output, price, and access is different in kind from applying regulatory pressure to other sectors. When a factory closes due to regulatory non-compliance, workers lose jobs and consumers pay more for a product. When a region’s agricultural capacity declines due to poorly designed policy, people go hungry.
The European Union’s Farm to Fork strategy, adopted in 2020, proposed reducing synthetic pesticide use by 50 percent and synthetic fertilizer use by 20 percent, while increasing organic farmland to 25 percent of total agricultural area, all by 2030.25 These are admirable environmental goals. But a 2021 analysis by Beckman and colleagues at the USDA Economic Research Service found that full implementation of the Farm to Fork targets would reduce EU agricultural output by 7 to 12 percent and increase consumer food prices by 5 to 11 percent.26 A JRC (Joint Research Centre of the European Commission) report from the same year found that global adoption of Farm to Fork-style policies would actually increase GHG emissions by up to 6 percent, because production displaced from Europe would move to regions with less efficient farming systems and weaker environmental controls.27
Agricultural environmental policy is essential; so is designing it carefully, with quantitative impact assessment, realistic timelines, and protections for the most vulnerable consumers.
What actually reduces food system emissions
The research literature on food system decarbonization converges on a consistent set of effective interventions, none of which involve dismantling existing agricultural production:
Reducing food waste. A 30 percent reduction in food loss and waste globally would reduce food system GHG emissions by roughly 8 to 10 percent.19 This is achievable through infrastructure investment (cold chains in developing countries), behavioral change (consumer education in wealthy ones), and regulatory reform (relaxing cosmetic standards for produce that create waste at the retail level).
Sustainable diets in high-income countries with a smart mix of protein sources, including poultry, pork, legumes, and dairy. Agriculture systems, including livestock production, should indeed operate at the lowest emissions level possible and with reduced antibiotic use to protect the environment, animals, and ultimately humans.
Improving agricultural productivity in low-income countries, particularly sub-Saharan Africa. Smil notes that average nitrogen application rates in sub-Saharan Africa are approximately 3 kilograms per hectare, compared to 50 kilograms in China and 30 kilograms in Europe.1 Increasing yields in Africa to levels achievable with modest fertilizer application and better seed varieties would allow the same food output from less land, reducing pressure on forests and biodiversity.
Improving nitrogen use efficiency in high-input farming systems through the technologies described earlier in the article.
None of these interventions require a technological revolution. They require investment, policy reform, and the political will to treat food production as the strategic priority it is.
References
1. Smil, V. (2024). How to Feed the World. Viking/Penguin Random House. (US edition 2025.)
2. Labrousse, E. (1944). La crise de l’économie française à la fin de l’Ancien Régime et au début de la Révolution. Presses Universitaires de France. Cited in McPhee, P. (2012). Liberty or Death: The French Revolution. Yale University Press.
3. Lagi, M., Bertrand, K.Z., & Bar-Yam, Y. (2011). The food crises and political instability in North Africa and the Middle East. New England Complex Systems Institute Preprint, arXiv:1108.2455. Available at: https://arxiv.org/abs/1108.2455
4. Bellemare, M.F. (2015). Rising food prices, food price volatility, and social unrest. American Journal of Agricultural Economics, 97(1), 1–21. https://doi.org/10.1093/ajae/aau038
5. FAO, IFAD, UNICEF, WFP and WHO (2024). The State of Food Security and Nutrition in the World 2024. FAO. https://www.fao.org/publications/sofi/2024/
6. Crippa, M., Solazzo, E., Guizzardi, D., Monforti-Ferrario, F., Tubiello, F.N., & Leip, A. (2021). Food systems are responsible for a third of global anthropogenic GHG emissions. Nature Food, 2, 198–209. https://doi.org/10.1038/s43016-021-00225-9
7. Gerber, P.J., Steinfeld, H., Henderson, B., Mottet, A., Opio, C., Dijkman, J., Falcucci, A., & Tempio, G. (2013). Tackling Climate Change Through Livestock: A Global Assessment of Emissions and Mitigation Opportunities. FAO. https://www.fao.org/3/i3437e/i3437e.pdf
8. Seufert, V., Ramankutty, N., & Foley, J.A. (2012). Comparing the yields of organic and conventional agriculture. Nature, 485, 229–232. https://doi.org/10.1038/nature11069
9. Lesur-Dumoulin, C., Malézieux, E., Ben-Ari, T., Langlais, C., & Makowski, D. (2017). Lower average yields but similar yield variability in organic versus conventional horticulture: a meta-analysis. Agronomy for Sustainable Development, 37, 45. https://doi.org/10.1007/s13593-017-0455-5
10. Ponisio, L.C., M’Gonigle, L.K., Mace, K.C., Palomino, J., de Valpine, P., & Kremen, C. (2015). Diversification practices reduce organic to conventional yield gap. Proceedings of the Royal Society B, 282, 20141396. https://doi.org/10.1098/rspb.2014.1396
11. Erisman, J.W., Sutton, M.A., Galloway, J., Klimont, Z., & Winiwarter, W. (2008). How a century of ammonia synthesis changed the world. Nature Geoscience, 1, 636–639. https://doi.org/10.1038/ngeo325
12. Risner, D., Kim, Y., Nguyen, D., Simons, C.W., & Spang, E. (2023). Preliminary techno-economic assessment of animal cell-based meat. bioRxiv. https://10.1101/2023.04.21.537778
13. Lynch, J., & Pierrehumbert, R. (2019). Climate impacts of cultured meat and beef cattle. Frontiers in Sustainable Food Systems, 3, 5. https://doi.org/10.3389/fsufs.2019.00005
14. Poore, J., & Nemecek, T. (2018). Reducing food’s environmental impacts through producers and consumers. Science, 360(6392), 987–992. https://doi.org/10.1126/science.aaq0216
15. Mekonnen, M.M., & Hoekstra, A.Y. (2010). The green, blue and grey water footprint of crops and derived crop products. Hydrology and Earth System Sciences, 15, 1577–1600. https://doi.org/10.5194/hess-15-1577-2011
16. Carrasco, L.R., Papworth, S.K., Reed, J., et al. (2017). High trade-offs between local and global demand for avocados. Nature Plants, 3, 1–3. See also Kibria, M.G., & Behrooz, M. (2022). Water footprint and environmental impact of avocado production. Sustainability, 14(2), 888.
17. Mottet, A., de Haan, C., Falcucci, A., Tempio, G., Opio, C., & Gerber, P. (2017). Livestock: On our plates or eating at our table? A new analysis of the feed/food debate. Global Food Security, 14, 1–8. https://doi.org/10.1016/j.gfs.2017.01.001
18. FAO (2011). Global Food Losses and Food Waste: Extent, Causes and Prevention. FAO. https://www.fao.org/3/mb060e/mb060e00.htm
19. Intergovernmental Panel on Climate Change (IPCC) (2019). Special Report on Climate Change and Land (SRCCL). Chapter 5: Food Security. https://www.ipcc.ch/srccl/chapter/chapter-5/
20. Zhang, X., Davidson, E.A., Mauzerall, D.L., Searchinger, T.D., Dumas, P., & Shen, Y. (2015). Managing nitrogen for sustainable development. Nature, 528, 51–59. https://doi.org/10.1038/nature15743
21. Cassman, K.G., Dobermann, A., & Walters, D.T. (2002). Agroecosystems, nitrogen-use efficiency, and nitrogen management. AMBIO: A Journal of the Human Environment, 31(2), 132–140.
22. Robertson, G.P., & Vitousek, P.M. (2009). Nitrogen in agriculture: Balancing the cost of an essential resource. Annual Review of Environment and Resources, 34, 97–125. https://doi.org/10.1146/annurev.environ.032108.105046
23. Springmann, M., Clark, M., Mason-D’Croz, D., Wiebe, K., Bodirsky, B.L., Lassaletta, L., de Vries, W., Vermeulen, S.J., Herrero, M., Carlson, K.M., Jonell, M., Troell, M., DeClerck, F., Gordon, L.J., Zurayk, R., Scarborough, P., Rayner, M., Loken, B., Fanzo, J., Godfray, H.C.J., Tilman, D., Rockstrom, J., & Willett, W. (2018). Options for keeping the food system within environmental limits. Nature, 562, 519–525. https://doi.org/10.1038/s41586-018-0594-0
24. United Nations, Department of Economic and Social Affairs (2022). World Population Prospects 2022. UN DESA. https://population.un.org/wpp/
25. European Commission (2020). Farm to Fork Strategy: For a Fair, Healthy and Environmentally-Friendly Food System. COM(2020) 381 final. https://ec.europa.eu/food/horizontal-topics/farm-fork-strategy_en
26. Beckman, J., Ivanic, M., Jelliffe, J.L., Burfisher, M.E., & Scott, S.M. (2020). Economic and Food Security Impacts of Agricultural Input Reduction Under the European Union Green Deal’s Farm to Fork and Biodiversity Strategies. USDA Economic Research Report EIB-30.
27. Barreiro-Hurle, J., Bogonos, M., Himics, M., Hristov, J., Pérez-Domínguez, I., Sahoo, A., Salputra, G., Weiss, F., Baldoni, E., and Elleby, C. (2021). Modelling environmental and climate ambition in the agricultural sector with the CAPRI model. JRC Technical Report EUR 30317 EN.
28. IPCC (2021). Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA. https://10.1017/9781009157896
29. Estévez, M., & Rui Alves Soares, C. (2025). Nutrient equivalence of plant-based and cultured meat: Gaps, bioavailability, and health perspectives. Nutrients, 17(24), 3860. https://doi.org/10.3390/nu17243860
30. Springmann, M., Godfray, H.C.J., Rayner, M., & Scarborough, P. (2016). Analysis and valuation of the health and climate change cobenefits of dietary change. Proceedings of the National Academy of Sciences, 113(15), 4146–4151. https://doi.org/10.1073/pnas.1523119113
31. Springmann, M., Wiebe, K., Mason-D’Croz, D., Sulser, T.B., Rayner, M., & Scarborough, P. (2018). Health and nutritional aspects of sustainable diet strategies and their association with environmental impacts: a global modelling analysis with country-level detail. Lancet Planetary Health, 2(10), e451–e461. https://doi.org/10.1016/S2542-5196(18)30206-7
32. World Bank (2025). Commodity Markets Price Data (The Pink Sheet), December 2025. World Bank Group. https://thedocs.worldbank.org/en/doc/18675f1d1639c7a34d463f59263ba0a2-0050012025/related/CMO-Pink-Sheet-December-2025.pdf
33. USDA Foreign Agricultural Service (2026). World Agricultural Production, April 2026. United States Department of Agriculture. https://apps.fas.usda.gov/psdonline/circulars/production.pdf
34. Counterpoint Research (2025). Global Smartphone Revenues Resume Growth in 2024 After Two Years, ASP Hits Record High, January 31, 2025. https://counterpointresearch.com/en/insights/global-smartphone-market-2024






 .
.