Secondary plant compounds against antibiotic-resistant E. coli

Due to incorrect therapeutic or preventive use of antibiotics in animal production as well as in human medicine, occurrence of antibiotic resistant pathogens has become a widespread problem. Enterobacteria in particular (e.g. Salmonella, Klebsiella, E. coli) possess a special mechanism of resistance. By producing special enzymes (ß-lactamases), they are able to withstand the attack of so-called ß-lactam antibiotics. The genes for this ability (resistance genes) can also be transferred to other bacteria resulting in a continuously increasing problem. Divers point mutations within the ß-lactamase genes lead to the occurrence of „Extended-Spectrum-Beta-Lactamases“ (ESBL), which are able to hydrolyse most of the ß-Lactam-antibiotics. AmpC Beta-Lactamases (AmpC) are enzymes, which express a resistance against penicillins, cephalosporins of the second and third generation as well as cephamycins.
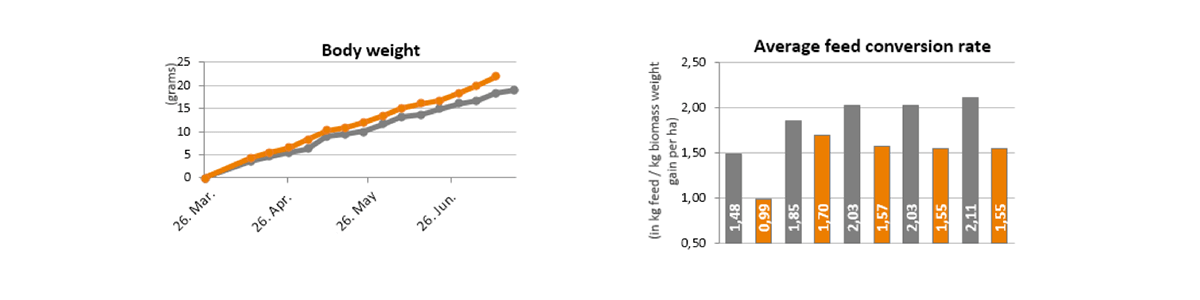
What are ß-lactam antibiotics?
The group of ß-lactam antibiotics consists of penicillins, cephalosporins, monobactams and carbapenems. A characteristic of these antibiotics is the lactam ring (marked in orange):
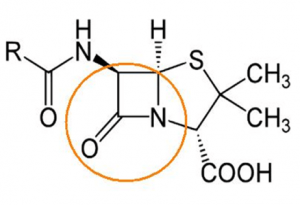
Mode of action of ß-lactam antibiotic
If a bacterial cell is growing, the cell wall also has to grow. For this purpose, existing conjunctions are cracked and new components are inserted. ß-lactam-antibiotics disturb the process of cell wall construction by blocking an enzyme needed, the transpeptidase. If crosslinks necessary for the stability of the cell wall cannot be created, the bacteria cannot survive. Resistant bacteria, which are able to produce ß-lactamases, destroy the ß-lactam antibiotics and prevent their own destruction.
Secondary plant compounds
Secondary plant compounds and their components are able to prevent or slow down the growth of moulds, yeasts, viruses and bacteria. They attack at various sites, particularly the membrane and the cytoplasm. Sometimes they change the whole morphology of the cell. In the case of gram-negative bacteria, secondary plant compounds (hydrophobic) have to be mixed with an emulsifier so that they can pass the cell wall which is open only for small hydrophilic solutes. The modes of action of secondary plant compounds depend on their chemical composition. It also depends on whether single substances or blends (with possible positive or negative synergies) are used. It has been observed that extracts of spices have a lower antimicrobial efficacy than the entire spice.
The best explained mode of action is the one of thymol and carvacrol, the major components of the oils of thyme and oregano. They are able to incorporate into the bacterial membrane and to disrupt its integrity. This increases the permeability of the cell membrane for ions and other small molecules such as ATP leading to the decrease of the electrochemical gradient above the cell membrane and to the loss of energy equivalents of the cell.
Trial (Scotland)
Design
Two strains of ESBL-producing and AmpC respectively, isolated from the field, a non-resistant strain of E. coli as control. Suspensions of the strains with 1×104 KBE/ml were incubated for 6-7 h at 37°C together with different concentrations of Activo Liquid or with cefotaxime, a cephalosporin. The suspensions were then put on LB-Agar plates and bacteria colonies were counted after a further 18-22h incubation at 37°C. Evaluation of the effects of Activo Liquid on ESBL-producing as well as on E. coli resistant for aminopenicillin and cephalosporin (AmpC)
Results
The antimicrobial efficacy of the blend of secondary plant compounds depended on concentration with bactericidal effect at higher concentrations and bacteriostatic at dilutions up to 0,1%. It is also possible that bacteria could develop a resistance to secondary plant compounds; the probability is however relatively low, due to the fact that essential oils contain hundreds of chemical components (more than antibiotics) making it difficult for bacteria to adapt.